FTIR Spectroscopy for Organic Pollutant Identification: Principles, Methods, and Advanced Applications
This article provides a comprehensive overview of Fourier Transform Infrared (FTIR) spectroscopy as a powerful tool for identifying and characterizing organic pollutants.
FTIR Spectroscopy for Organic Pollutant Identification: Principles, Methods, and Advanced Applications
Abstract
This article provides a comprehensive overview of Fourier Transform Infrared (FTIR) spectroscopy as a powerful tool for identifying and characterizing organic pollutants. Tailored for researchers and drug development professionals, it covers the foundational principles of molecular vibration and spectral interpretation, detailed methodologies for sample preparation and analysis across various matrices, strategies to overcome common analytical challenges, and the validation of FTIR through integration with complementary techniques. The content synthesizes current applications—from environmental microplastics to food safety and forensic analysis—and explores future directions, including the growing role of machine learning and portable instrumentation in advancing analytical capabilities.
The Fundamentals of FTIR: How Molecular Fingerprints Unmask Organic Pollutants
Fourier Transform Infrared (FTIR) spectroscopy is a powerful analytical technique that identifies organic pollutants by detecting their unique molecular fingerprints. It operates on a fundamental principle: when molecules are exposed to infrared light, their chemical bonds vibrate at specific frequencies, absorbing light energy in the process. By measuring these absorption patterns, FTIR provides a non-destructive and highly specific method for determining the molecular composition of a sample, making it indispensable in environmental research for tracking and identifying contaminants [1] [2].
Core Principles: The Interaction of Light and Matter
The operational principle of FTIR is based on the fact that chemical bonds in a molecule are not static; they behave like springs, constantly vibrating. When infrared light, which shares the same energy range as these molecular vibrations, interacts with a sample, bonds can absorb specific wavelengths of light, leading to changes in their vibrational energy states [2].
Molecular Vibrations and Infrared Absorption
The frequency of a bond's vibration depends on the masses of the atoms involved and the strength of the bond between them. Lighter atoms and stronger bonds vibrate at higher frequencies [2]. These vibrations are quantized, meaning molecules will only absorb infrared light at frequencies that match their natural vibrational modes. The primary types of vibrations are:
- Stretching: A rhythmic change in the interatomic distance along the bond axis.
- Bending: A change in the bond angle between atoms.
The resulting infrared absorption spectrum acts as a molecular fingerprint, unique to the chemical structure of the substance being analyzed [2].
From Interferogram to Spectrum: The Role of the Fourier Transform
Unlike older dispersive spectrometers, FTIR instruments use an interferometer to measure all infrared frequencies simultaneously. The core components and process are as follows [1] [2]:
- Infrared Source: A glowing filament emits a broad beam of infrared light.
- Interferometer: The heart of the system. The beam is split into two paths—one reflecting off a fixed mirror and the other off a moving mirror.
- Sample Interaction: The recombined beams pass through or reflect off the sample, where specific frequencies are absorbed.
- Detection: The detector records a complex signal called an interferogram, which encodes intensity information from all wavelengths as a function of the moving mirror's position.
- Fourier Transform: A mathematical algorithm processes the interferogram, converting it from a time-domain signal into a familiar frequency-domain spectrum.
This process, summarized in the diagram below, gives FTIR significant advantages in speed, sensitivity, and signal-to-noise ratio.
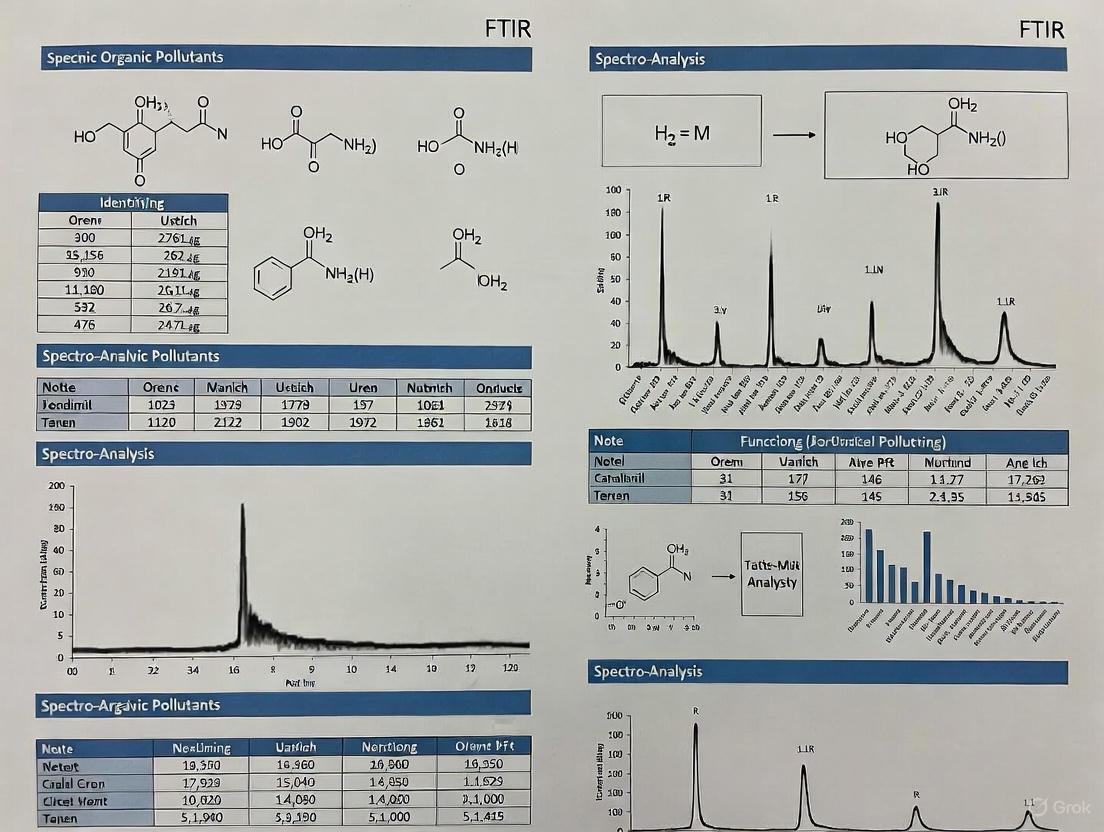
FTIR Spectral Analysis for Organic Pollutants
The final output of an FTIR analysis is a spectrum plotting absorbance (or transmittance) against wavenumber (cm⁻¹). The peaks, or absorption bands, correspond to the vibrational modes of specific functional groups within the molecules, allowing for definitive identification [1] [2].
Characteristic Functional Group Absorptions
The table below lists the characteristic infrared absorption bands for functional groups commonly found in organic pollutants.
Table 1: Characteristic IR Absorptions of Key Functional Groups
| Functional Group | Bond | Typical Absorption Range (cm⁻¹) | Vibration Type |
|---|---|---|---|
| Hydroxyl | O-H | 3200 - 3600 (broad) | Stretching [2] |
| Carbonyl | C=O | 1690 - 1810 | Stretching [3] |
| Alkenyl | C=C | 1600 - 1680 | Stretching [3] |
| Carbon-Oxygen | C-O | 1000 - 1300 | Stretching |
Spectral Interpretation and Degradation Assessment
In environmental analysis, FTIR not only identifies pollutants but can also assess their degradation state. For instance, in microplastic (MP) pollution research, weathering indexes are calculated from the spectrum to track polymer aging [3]:
- Carbonyl Index (CI): Measures the formation of carbonyl groups (C=O) due to photo-oxidation, calculated as the ratio of absorbance around 1710 cm⁻¹ to a reference peak.
- Hydroxyl Index (HI): Tracks the formation of hydroxyl groups (O-H), indicating oxidation.
- Carbon-Oxygen Index (COI): Reflects changes in C-O bond vibrations.
These indices have been crucial in studying MPs in freshwater environments, showing that particles degraded in natural settings have unique spectral properties compared to those aged artificially in labs [3].
Experimental Protocols for Pollutant Analysis
Sample Preparation and Analysis
The choice of protocol depends on the sample's physical state and nature.
- Attenuated Total Reflectance (ATR): This is now a prevalent method for solid and liquid samples. It requires minimal preparation: a small amount of the sample is placed on a crystal (e.g., diamond), and pressure is applied to ensure good contact for analysis. It is ideal for identifying microplastics collected from water or soil [3] [4].
- Transmission: The sample is embedded in a KBr pellet or dissolved in a solvent and placed in a cell for analysis [1].
- Gas Cell: For analyzing gaseous emissions, the sample is introduced into an evacuated cell with a long path length to enhance sensitivity, as prescribed in methods like EPA Method 320 for monitoring stack emissions [5].
A Workflow for Microplastic Identification in Environmental Samples
The following diagram outlines a typical FTIR workflow for identifying and characterizing microplastics in a river water sample, integrating both chemical identification and degradation assessment.
The Scientist's Toolkit: Essential Reagents and Materials
Successful FTIR analysis relies on a set of key materials and reagents, each serving a specific function in sample preparation and instrument operation.
Table 2: Key Research Reagent Solutions and Materials
| Item | Function in FTIR Analysis | Example Use-Case |
|---|---|---|
| ATR Crystal (Diamond) | Provides a durable, chemically inert surface for internal reflection of IR light for solid and liquid samples. | Analysis of microplastic particles [3] and ambient aerosols [6]. |
| Potassium Bromide (KBr) | A transparent medium in the IR region used to prepare pellets for transmission analysis of solid samples. | Traditional method for analyzing powdered soils or polymers [1]. |
| High-Purity Solvents | Used to dissolve or dilute samples for liquid cell analysis; must be IR-transparent in regions of interest. | Preparing solutions of organic contaminants for quantitative analysis. |
| Calibration Gas Mixtures | Certified standard gases used to validate and calibrate the instrument for quantitative gas analysis. | Compliance testing of industrial emissions as per EPA Method 320 [5]. |
| Spectral Libraries | Databases of known compound spectra used as a reference for identifying unknown materials in a sample. | Confirming the identity of a seized narcotic or an unknown polymer [7]. |
Applications in Organic Pollutant Research
The versatility of FTIR makes it a cornerstone technique across numerous environmental applications.
- Microplastic Pollution: FTIR is the gold standard for identifying the polymer type (e.g., polyethylene, polypropylene) of microplastics found in water, soil, and even insects, revealing pathways of contamination through ecosystems [3] [4].
- Air Quality and Aerosols: Using ATR-FTIR, researchers can characterize the organic functional groups and inorganic ions in ambient aerosols, helping to identify pollution sources and their impact on air quality in protected regions [6].
- Regulatory Compliance: The U.S. Environmental Protection Agency (EPA) has standardized FTIR for environmental monitoring. EPA Method 320 is an extractive FTIR procedure for measuring vapor-phase organic and inorganic emissions from stationary sources, underscoring the technique's reliability for regulatory compliance [5].
- Toxic Metal Interactions: While FTIR does not directly detect elemental metals, it is proficient at identifying functional groups that bind to toxic metals (e.g., As, Pb, Cd) in food and environmental matrices. This helps in understanding the mechanisms of metal bioavailability and toxicity [8].
Comparative Techniques and Future Outlook
While other methods like Atomic Absorption Spectroscopy (AAS) and Inductively Coupled Plasma Mass Spectrometry (ICP-MS) offer lower detection limits for elemental analysis, they are often more time-consuming and costly, and do not provide molecular-level information [8]. FTIR's strength lies in its rapid, non-destructive provision of a molecular fingerprint.
Future directions in FTIR analysis involve greater integration with chemometric models and hybrid analytical systems to improve detection limits and analytical precision [8]. The development of more portable and imaging-based FTIR systems also promises real-time, in-field monitoring of organic pollutants, further solidifying its role as an essential tool in environmental science.
Fourier Transform Infrared (FTIR) spectroscopy has emerged as an indispensable analytical technique in environmental science for identifying and characterizing organic pollutants. This technology operates on the principle that molecules absorb specific frequencies of infrared light corresponding to the vibrational frequencies of their chemical bonds, creating a unique molecular "fingerprint" that can be used for precise identification [9]. The resulting spectrum provides a detailed profile of the functional groups and molecular structures present in a sample, enabling researchers to detect everything from microplastics in freshwater systems to volatile organic compounds (VOCs) in air and toxic metals in food matrices [3] [8] [10]. For environmental researchers investigating organic pollutants, FTIR offers a powerful combination of specificity, sensitivity, and versatility, allowing for the rapid screening of contaminants across diverse environmental samples including solids, liquids, and gases [1] [11].
The application of FTIR in pollution research has gained significant momentum in recent years, particularly in the study of complex environmental challenges such as microplastic pollution, where it is used to determine both the chemical composition and the degree of ageing of plastic fragments [3]. Furthermore, advanced FTIR methodologies, including integration with chemometric models and hybrid analytical systems, have markedly enhanced the sensitivity, resolution, and specificity of pollution monitoring techniques [8]. This guide provides a comprehensive technical framework for interpreting FTIR spectra within the context of organic pollutant research, detailing characteristic functional groups, experimental protocols, and advanced applications relevant to environmental scientists.
Fundamental Principles of FTIR Spectral Interpretation
The FTIR Spectrum and Its Regions
An FTIR spectrum is a two-dimensional graph representing the intensity of infrared light absorbed by a sample across a range of frequencies, typically expressed in wavenumbers (cm⁻¹) [9]. The mid-infrared region (4,000–400 cm⁻¹) is the most analytically useful for identifying organic functional groups and is divided into four key regions, each associated with specific types of molecular vibrations [9]:
- The Single Bond Region (2,500–4,000 cm⁻¹): Dominated by stretching vibrations of O-H, N-H, and C-H bonds.
- The Triple Bond Region (2,000–2,500 cm⁻¹): Characteristic of triple bond stretching vibrations such as C≡C and C≡N.
- The Double Bond Region (1,500–2,000 cm⁻¹): Features important carbonyl (C=O) stretching vibrations and C=C double bonds.
- The Fingerprint Region (600–1,500 cm⁻¹): Contains complex patterns arising from bending vibrations and single bond stretches, providing a unique identifier for specific compounds.
For microbiological and environmental samples, researchers often utilize five specific spectral windows within the mid-IR spectrum to focus on particular biological or chemical constituents [9]. These windows help in classifying and interpreting complex spectral data from environmental samples.
Characteristic Absorption Bands of Common Organic Pollutants
The identification of organic pollutants relies on recognizing characteristic absorption bands of functional groups. The table below summarizes key vibrational frequencies for functional groups commonly found in environmental contaminants:
Table 1: Characteristic FTIR Absorption Frequencies of Common Organic Functional Groups in Pollutants
| Functional Group | Bond Type | Vibration Mode | Frequency Range (cm⁻¹) | Pollutant Examples |
|---|---|---|---|---|
| Hydroxyl Group | O-H | Stretching | 3,200–3,600 [9] | Alcohols, phenols |
| Carbonyl Group | C=O | Stretching | 1,690–1,810 [3] | Ketones, aldehydes (1,680–1,750 [9]), carboxylic acids (1,700–1,750 [9]) |
| Aromatic Ring | C=C | Stretching | 1,450–1,600 [9] | Polystyrene, PAHs |
| Methylene Group | C-H | Stretching (Alkane) | 2,850–2,960 [9] | Polyethylene, polypropylene |
| Amine Group | N-H | Stretching | 3,300–3,500 [9] | Azo dyes, pharmaceuticals |
| Carbon-Oxygen Bond | C-O | Stretching | 1,000–1,300 [3] | Alcohols, carboxylic acids, esters |
The carbonyl group (C=O) is particularly significant in environmental degradation studies. For instance, in microplastic research, the formation of carbonyl groups on polymer surfaces upon UV exposure is a key indicator of photodegradation, which can be tracked using the Carbonyl Index [3]. Similarly, the hydroxyl index (O-H stretching between 3,100–3,700 cm⁻¹) provides insights into the extent of weathering in environmental plastic particles [3].
Experimental Protocols for Pollutant Analysis
Sample Preparation and Measurement Techniques
The choice of sampling methodology is critical for obtaining high-quality FTIR spectra and depends on the physical state and nature of the environmental sample.
Attenuated Total Reflectance (ATR) for Solid Samples ATR-FTIR has become a mainstream technique for analyzing solid environmental samples such as microplastics, soil, and aerosols [3] [6]. This method requires minimal sample preparation and is ideal for irregularly shaped materials.
- Procedure: A small sample is placed in direct contact with a high-refractive-index crystal (e.g., diamond). Infrared light undergoes total internal reflection within the crystal, generating an evanescent wave that penetrates the sample, typically 0.5–2 microns deep, and is absorbed at characteristic frequencies [3].
- Applications: Identification of polymer types in microplastics [3] [12], characterization of organic functional groups and inorganic ions in ambient aerosols [6], and analysis of contaminated soils [4].
- Protocol Note: For quantitative analysis of aerosols, a method utilizing ATR-FTIR with multivariate calibration requires only small sample sizes and no preparation, making it suitable for low-concentration environments like national parks [6].
Transmission Cells for Gas Analysis FTIR gas analysis is employed for monitoring volatile organic compounds (VOCs) in air and industrial emissions [10] [11].
- Procedure: Gas samples are contained in a cell with infrared-transparent windows (e.g., KBr). The infrared beam passes through the sample, and the spectrum is collected. For increased sensitivity, multipass cells with extended path lengths (e.g., 10 meters) are used to detect VOCs down to parts-per-million (ppm) levels [10].
- Applications: Monitoring of VOCs like styrene, acetone, and ethanol in indoor air [10]; analysis of engine exhaust emissions [11].
- Protocol Note: Experiments calibrating FTIR for VOC quantification involve introducing known amounts of liquid VOC into an evaporation chamber, allowing it to equilibrate, and then transferring the vapor into a multipass cell for analysis, with concentrations simultaneously monitored by a Photo-Ionization Detector (PID) sensor [10].
Liquid Analysis for Water Contaminants Water samples can be analyzed directly or after filtration to concentrate pollutants.
- Procedure for Microplastics: Water is filtered onto specialized filters, and the collected particles are analyzed using micro-FTIR imaging for small particles or reflectance-FTIR for larger particles [11].
- Procedure for Dissolved Organics: A volume of 2–5 ml of water sample can be subjected directly to FTIR analysis to identify polymers and other organic contaminants [4].
Workflow for Pollutant Identification and Characterization
The following diagram illustrates the standard workflow for FTIR-based analysis of environmental pollutants, integrating sampling, measurement, and data interpretation steps:
Diagram 1: FTIR Analysis Workflow for Environmental Pollutants
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful FTIR analysis of environmental pollutants requires specific reagents, accessories, and analytical tools. The following table details key solutions and materials essential for research in this field:
Table 2: Essential Research Reagents and Materials for FTIR Analysis of Pollutants
| Item | Function/Application | Technical Specifications |
|---|---|---|
| ATR Crystals | Enables direct analysis of solid samples without preparation. | Diamond crystal plate for durability; wavenumber range 4000–500 cm⁻¹ [3]. |
| Multipass Gas Cells | Increases path length for enhanced sensitivity in gas analysis. | 10-meter path length, 2L volume for detection of VOCs at ppm levels [10]. |
| Infrared-Transparent Windows | Windows for gas and liquid cells. | KBr windows, 25mm wide, 4mm thick [10]. |
| Spectral Libraries | Automated identification of unknown polymers and pollutants. | Custom libraries for common environmental polymers (PE, PP, PS, PET) [12] [11]. |
| Calibrated VOC Standards | Quantitative analysis of volatile organic compounds in air. | Certified reference materials for styrene, acetone, ethanol, isopropanol [10]. |
| Photo-Ionization Detector (PID) | Real-time monitoring of VOC concentrations for FTIR calibration. | Sensitivity of 1 ppmv, used with correction factors for different VOCs [10]. |
| Motorized XY Stage & Camera | Semi-automated analysis of multiple particles (e.g., microplastics). | Movement accuracy ±0.001 mm, integrated imaging for particle recognition [12]. |
| Specialized Filters for Water Sampling | Collection and concentration of microplastics from water samples. | Used with aqueous sample filtration for subsequent FTIR imaging [11]. |
Advanced Applications in Environmental Research
Microplastic Pollution and Degradation Assessment
FTIR spectroscopy has become a cornerstone technique in microplastic pollution research, enabling both polymer identification and assessment of environmental degradation. The ageing of plastics is evaluated using specific spectral indexes that track the formation of oxidative functional groups:
- Carbonyl Index (CI): Measures the intensity of the carbonyl (C=O) stretching band (1690–1810 cm⁻¹) relative to a reference band, indicating photo-oxidative degradation [3]. For example, environmental polyethylene pellets from a freshwater river showed CI values ranging from 0.05 to 0.26 [3].
- Hydroxyl Index (HI): Based on the O-H stretching region (3100–3700 cm⁻¹), indicating the formation of hydroxyl, carboxyl, or phenol groups during weathering [3].
- Carbon-Oxygen Index (COI): Related to vibrations of C-O bonds found in carbohydrates, alkanes, secondary alcohols, and ketones [3].
Recent advancements include the development of semi-automated systems like the Microplastic Analyzer using Reflectance-FTIR Semi-automatically (MARS), which integrates a motorized stage, cameras, and reflectance-FTIR to rapidly identify the number, size, and polymer type of large microplastics with over 98% accuracy compared to conventional ATR-FTIR [12].
Monitoring of Volatile Organic Compounds (VOCs) in Air
FTIR spectroscopy provides a powerful tool for quantitative monitoring of hazardous VOCs in both indoor and outdoor environments. The technique's selectivity allows for discrimination between different chemical agents, which is a limitation of many conventional sensors [10]. Key application areas include:
- Occupational Health Monitoring: Real-time detection of styrene, acetone, ethanol, and isopropanol in industrial settings [10].
- Indoor Air Quality Assessment: Identification and quantification of VOCs in working and living environments where these compounds pose serious health risks [10].
- Source Emissions Analysis: Continuous monitoring of stack emissions from various industrial processes, providing real-time data on hundreds of compounds simultaneously without need for calibration [11].
The high sensitivity achieved with multipass cells (path lengths of 10 meters) enables detection down to part-per-million (ppm) levels, making FTIR suitable for compliance monitoring with occupational exposure limits [10].
Integrated Environmental Monitoring
FTIR spectroscopy facilitates comprehensive environmental assessment by analyzing pollutants across multiple interconnected compartments:
- Soil-Vater-Insect Systems: Research has demonstrated the transfer of microplastics between environmental compartments, with FTIR analysis confirming the presence of polypropylene/polystyrene (PP/PS), polyethylene (PE), polyethylene terephthalate (PET), and polyamide (PA) in soil, water, and insect samples [4]. Insect samples, particularly beetles and bees, showed high microplastic adherence, suggesting their role as vectors for microplastic dissemination.
- Atmospheric Aerosols: ATR-FTIR characterization of organic functional groups and inorganic ions in ambient aerosols at rural sites helps identify pollution sources and transport patterns [6].
- Food Safety: FTIR is increasingly used for profiling toxic metals and organic contaminants in food matrices, addressing serious public health challenges related to food contamination [8].
FTIR spectroscopy provides environmental researchers with a powerful analytical platform for identifying and characterizing organic pollutants across diverse environmental matrices. The technique's ability to detect specific functional groups and provide molecular fingerprints makes it indispensable for monitoring contaminants ranging from microplastics in freshwater systems to VOCs in atmospheric samples. As environmental challenges grow more complex, ongoing advancements in FTIR technology—including semi-automated analysis systems, integration with chemometric models, and the development of portable field-deployable instruments—promise to further enhance our capability to monitor, understand, and mitigate the impacts of organic pollutants on ecosystems and human health. The continued refinement of spectral libraries and quantitative methodologies will ensure FTIR remains at the forefront of environmental analytical science, providing critical data to support evidence-based environmental management and policy decisions.
Fourier Transform Infrared (FTIR) spectroscopy has emerged as a powerful analytical technique for identifying molecular compounds, playing a critical role in environmental monitoring, pharmaceutical development, and materials science. This non-destructive method provides a unique molecular fingerprint by measuring a sample's absorption of infrared light at various wavelengths, enabling researchers to identify functional groups and molecular structures present in a sample [13]. Within environmental research, FTIR has become particularly valuable for detecting and characterizing organic pollutants across diverse matrices, offering rapid, cost-effective analysis with minimal sample preparation [8] [14]. This guide details the comprehensive analytical workflow from sample collection to spectral interpretation, providing researchers with standardized protocols for reliable organic pollutant identification.
Fundamental Principles of FTIR Spectroscopy
FTIR spectroscopy operates on the principle that molecules absorb specific frequencies of infrared radiation corresponding to their natural vibrational frequencies. When infrared radiation is applied to a sample, covalent bonds within the molecules undergo rotational, bending, and stretching vibrations, absorbing energy at characteristic frequencies [15]. The resulting spectrum plots these absorptions, creating a distinctive pattern that serves as a molecular fingerprint for identification purposes [16].
The core components of an FTIR spectrometer include an infrared source, interferometer, sample chamber, detector, and computer system. The interferometer creates a beam of infrared light with a known wavelength range, which passes through or reflects off the sample. The detector measures the intensity of the transmitted or reflected light as a function of its wavelength, and a Fourier transform algorithm converts this raw data into a recognizable infrared spectrum [13]. This process enables the precise molecular characterization of organic and inorganic materials, making FTIR indispensable for contemporary analytical research.
Sample Collection and Preparation
Collection Protocols
Proper sample collection is foundational to obtaining reliable FTIR results. For organic pollutant analysis, collection methods vary significantly based on the matrix:
- Water Samples: For microplastics analysis in drinking water, recent meta-analyses recommend collecting sufficient volume (typically 1-10 liters depending on expected contaminant load) using clean, non-plastic containers to avoid cross-contamination [17]. Sampling locations should be selected based on potential pollution sources and hydrodynamic considerations.
- Solid Samples: Soil, sediment, or biological tissues require representative sampling using stainless steel tools. For heterogeneous materials, composite sampling from multiple locations within the target area ensures representative analysis [8].
- Airborne Particulates: Air sampling for gaseous or particulate pollutants employs specialized filtration systems or impingers, with careful attention to flow rates, collection times, and potential artifact formation [14].
Throughout collection, stringent contamination control measures are essential, including the use of field blanks, replication, and proper sample labeling with complete metadata documentation.
Preparation Techniques
Sample preparation must be tailored to the sample matrix and analytical objectives:
- Liquid Samples: Water samples often require filtration to concentrate particulate pollutants. Cellulose nitrate filters with 0.45 μm pore size have demonstrated superior retention capabilities for microplastics and are recommended for standardized protocols [17]. For dissolved organics, liquid-liquid extraction or solid-phase extraction may be necessary before analysis.
- Solid Samples: Soils, sediments, and biological tissues typically require homogenization and drying before analysis. For complex matrices, purification steps such as density separation (using zinc chloride or sodium iodide solutions) effectively separate organic pollutants from mineral components [17].
- Staining for Enhanced Detection: For fluorescent detection of polymers, Nile red staining (1 mg/L in methanol) applied to filtered samples significantly improves visualization and quantification capabilities. Samples are typically incubated at 30°C for 30 minutes after staining to optimize fluorescence [17].
Table 1: Recommended Filtration Materials for Different Sample Types
| Filter Material | Pore Size (μm) | Optimal Application | Key Advantages |
|---|---|---|---|
| Cellulose Nitrate | 0.45 | Microplastics in drinking water | Superior retention, compatibility with FTIR |
| Polycarbonate | 0.4 | Nanoparticle analysis | Smooth surface for particle inspection |
| Silicon | 0.5 | High-resolution imaging | IR transparency for transmission analysis |
| Aluminum Oxide | 0.2 | Small particle retention | Chemical resistance, high loading capacity |
Instrumental Analysis and Data Acquisition
FTIR Configuration
Modern FTIR spectrometers offer multiple sampling accessories tailored to different sample types:
- Attenuated Total Reflectance (ATR): This technique requires minimal sample preparation and is ideal for solid and liquid samples. ATR utilizes the phenomenon of total internal reflection, where the infrared beam penetrates a short distance into the sample in contact with the crystal, generating a high-quality spectrum with virtually no preparation [14].
- Transmission Spectroscopy: The traditional method where infrared light passes directly through a thin sample. This approach requires samples to be either thin enough to be transparent to IR light or embedded in IR-transparent matrices like potassium bromide (KBr) pellets [18].
- Diffuse Reflectance (DRIFTS): Particularly useful for powdered samples, DRIFTS collects and measures scattered infrared light from rough surface samples, requiring minimal preparation [18].
For organic pollutant analysis, the PerkinElmer Spectrum Two FTIR spectrometer configured with a deuterated triglycine sulfate (DTGS) detector provides excellent sensitivity across the 400-4000 cm⁻¹ range. Recommended parameters include:
- Spectral resolution: 1-4 cm⁻¹
- Number of scans: 8-32 (balancing signal-to-noise ratio with analysis time)
- Apodization function: Norton-Beer medium for favorable linearity [19]
Quality Assurance Protocols
Ensuring spectral quality is paramount for reliable interpretation. Key quality indicators include:
- Low noise (minimal "fuzz" in the baseline)
- Little or no baseline offset
- Flat baseline
- Peaks on scale (between 0 and 2 absorbance units)
- Freedom from spectral artifacts [18]
Common artifacts include water vapor (peaks near 3400 cm⁻¹ and 1600 cm⁻¹) and carbon dioxide (doublet near 2350 cm⁻¹), which can be minimized by purging the instrument with dry nitrogen and regularly running background spectra [18]. Regular instrument calibration using polystyrene standards verifies wavelength accuracy and resolution.
Diagram 1: FTIR Analytical Workflow. This flowchart outlines the systematic process from sample collection to final interpretation, with quality assessment as a critical gatekeeping step.
Spectral Interpretation and Data Analysis
Systematic Interpretation Approach
Effective FTIR spectral interpretation requires a structured methodology. The following 12-step process ensures comprehensive analysis:
- Verify Spectral Quality: Confirm the spectrum exhibits low noise, minimal baseline offset, and freedom from artifacts before interpretation [18].
- Avoid Mixtures When Possible: Complex mixtures complicate interpretation; separate components when feasible [18].
- Leverage Sample Knowledge: Utilize all available information about the sample's origin, properties, and expected composition [18].
- Document Measurement Conditions: Note instrumental resolution, sampling method, and any spectral processing applied [18].
- Identify Spectral Artifacts: Recognize and discount peaks from water vapor, CO₂, or sample preparation materials [18].
- Mark Known Components: Identify peaks from expected compounds or sample matrices before analyzing unknown features [18].
- Read Left to Right: Systematically examine the spectrum from high to low wavenumbers, noting presence/absence of key functional groups [18].
- Assign Intense Bands First: Prominent peaks are typically most diagnostically useful and should be identified initially [18].
- Locate Secondary Bands: Find less intense peaks associated with already-identified functional groups [18].
- Assign Remaining Bands: Address remaining unassigned peaks as needed for the specific analysis [18].
- Consult Reference Spectra: Compare unknown spectra against database references for verification [16].
- Corroborate with Complementary Techniques: Confirm interpretations with additional analytical methods when possible [18].
Characteristic Spectral Regions
FTIR spectra are typically divided into distinct regions, each associated with specific molecular vibrations:
Table 2: Characteristic FTIR Absorption Regions for Organic Pollutants
| Spectral Region (cm⁻¹) | Bond/Vibration Type | Representative Functional Groups | Pollutant Examples |
|---|---|---|---|
| 3600-3200 | O-H, N-H stretching | Alcohols, phenols, amines | Bisphenol A, aniline derivatives |
| 3100-2800 | C-H stretching | Alkanes, alkenes, aromatics | Petroleum hydrocarbons, PAHs |
| 2250-2100 | C≡C, C≡N stretching | Alkynes, nitriles | Acrylonitrile, propynyl compounds |
| 1850-1650 | C=O stretching | Ketones, aldehydes, esters | Phthalates, ketone solvents |
| 1650-1450 | C=C, N-H bending | Alkenes, aromatics, amines | Styrene, aniline, polyaromatics |
| 1300-1000 | C-O, C-N stretching | Alcohols, esters, amines | Plasticizers, surfactants |
| 1000-500 | Fingerprint region | Complex molecular vibrations | Polymer fingerprints, unique identifiers |
The region between 4000-1500 cm⁻¹ is particularly valuable for identifying specific functional groups, while the fingerprint region (1500-500 cm⁻¹) provides unique patterns that enable discrimination between structurally similar compounds [15]. For example, carbonyl (C=O) stretching in esters and ketones appears as a strong, sharp peak between 1750-1680 cm⁻¹, while O-H stretching in alcohols and carboxylic acids produces a broad band between 3650-2500 cm⁻¹ [16] [15].
Advanced Data Analysis Techniques
Modern FTIR analysis increasingly incorporates chemometric methods to extract maximum information from spectral data:
- Baseline Correction: The adaptive smoothness parameter penalized least squares (asPLS) method effectively corrects for baseline drift caused by environmental variations during spectral acquisition [19].
- Multivariate Analysis: Principal Component Analysis (PCA) and Partial Least Squares (PLS) regression enable classification of complex samples and quantitative analysis of multiple components simultaneously [14] [20].
- Spectral Variable Selection: For overlapping absorption peaks, wavelength selection methods based on variable impact and population analysis improve quantitative accuracy when coupled with backpropagation neural networks [19].
For quantitative analysis, the Correlation Analysis-Interval Random Frog-Partial Least Squares (CA-IRF-PLS) method has demonstrated superior performance by selecting optimal spectral bands specific to different analyte classes, significantly enhancing predictive accuracy for complex biological and environmental samples [20].
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Materials for FTIR Analysis of Organic Pollutants
| Item | Function | Application Notes |
|---|---|---|
| Cellulose Nitrate Filters (0.45 μm) | Sample filtration and concentration | Superior retention for microplastics in water samples [17] |
| Nile Red Stain | Fluorescent detection of polymers | Use at 1 mg/L in methanol with 30 min incubation at 30°C [17] |
| Potassium Bromide (KBr) | Pellet preparation for transmission analysis | IR-transparent matrix for solid samples |
| ATR Crystals (diamond, ZnSe) | Surface analysis with minimal preparation | Diamond: robust, chemical-resistant; ZnSe: wider spectral range |
| Deuterated Triglycine Sulfate (DTGS) Detector | Infrared radiation detection | Standard sensitivity for routine analysis |
| Polystyrene Standard | Instrument calibration | Verifies wavelength accuracy and resolution |
| Zinc Chloride Solution | Density separation | Isolates organic pollutants from mineral matrices |
| Nitrogen Purge System | Reduces atmospheric interference | Minimizes water vapor and CO₂ artifacts in spectra |
Applications in Organic Pollutant Research
FTIR spectroscopy has proven particularly valuable in several key areas of environmental pollution research:
- Microplastics Identification: Standardized FTIR methodologies enable reliable detection and classification of microplastics in drinking water, with cellulose nitrate filters and Nile red staining establishing robust protocols for comparative studies [17]. This approach has revealed concerning microplastic levels in various water sources, with one study finding 65.66% of water samples from storage tanks contained detectable microplastics [17].
- Toxic Metal-Organic Complexes: While FTIR does not directly quantify toxic metals, it proficiently identifies functional groups involved in metal binding, characterizing interactions between organic pollutants and metals like cadmium, lead, and mercury in environmental samples [8].
- Atmospheric Pollutants: FTIR gas analyzers demonstrate exceptional sensitivity for greenhouse gases and volatile organic compounds, with detection limits reaching 0.5 ppm for CH₄, 1 ppm for CO, and 0.2 ppm for C₂H₂ in complex mixtures [19].
- Clinical and Biological Monitoring: FTIR analysis of blood, saliva, and urine enables non-invasive detection of organic pollutants and their metabolic products, with pattern recognition algorithms successfully classifying exposure-related pathologies with high sensitivity and specificity (Rcv > 0.93) [14].
The comprehensive analytical workflow from sample to spectral data in FTIR spectroscopy provides researchers with a powerful methodology for organic pollutant identification and characterization. Each stage—from careful sample collection and appropriate preparation through optimized instrumental analysis to systematic spectral interpretation—contributes significantly to the reliability and accuracy of final results. The ongoing integration of advanced chemometric techniques with traditional spectral interpretation continues to expand FTIR applications in environmental monitoring, clinical diagnostics, and regulatory compliance. As standardization efforts progress and portable instrumentation becomes more sophisticated, FTIR spectroscopy is positioned to play an increasingly vital role in global environmental and public health protection through precise organic pollutant monitoring.
Fourier Transform Infrared (FTIR) spectroscopy has established itself as a cornerstone analytical technique in modern laboratories due to its remarkable versatility in analyzing matter across all physical states. This universal detection capability makes it indispensable for identifying organic pollutants, which frequently exist in complex environmental matrices as solids, liquids, and gases. The fundamental principle of FTIR involves measuring molecular vibrations through the absorption of infrared radiation, creating a unique molecular fingerprint for virtually any material [21]. Unlike techniques limited to specific sample types, FTIR's adaptability across physical states provides researchers with a unified analytical platform, eliminating the need for multiple instrumentation approaches and facilitating direct comparison of pollutants from different sources and phases.
The operational principle enabling this universal application centers on the interaction between infrared light and molecular bonds. When IR radiation interacts with a sample, specific frequencies are absorbed, corresponding to molecular bond vibrations such as stretching, bending, or twisting of dipoles [21]. These absorption patterns are highly characteristic of specific functional groups and molecular structures, allowing for both qualitative identification and quantitative measurement. The Fourier transform algorithm converts raw interferogram data into meaningful spectra by deconvoluting the signal from all wavelengths measured simultaneously, providing the speed and sensitivity necessary for analyzing diverse sample types [21] [22]. For organic pollutant research, this means that the same instrumental platform can identify hydrocarbon contaminants in soil (solids), pesticides in water (liquids), and volatile organic compounds in air (gases), creating a comprehensive analytical approach to environmental monitoring.
Core Principles and Instrumentation
Fundamental Operating Principles
FTIR spectroscopy operates on the principle that molecules absorb specific frequencies of infrared radiation that correspond to their natural vibrational frequencies. These vibrations occur only when the incident IR radiation matches the energy difference between vibrational ground and excited states, and when the interaction causes a change in the dipole moment of the molecule [22]. The resulting absorption spectrum provides a characteristic molecular fingerprint spanning the mid-infrared region (4000-400 cm⁻¹), where most fundamental molecular vibrations occur [21] [22]. The Fourier transform advantage arises from the simultaneous measurement of all wavelengths via an interferometer, most commonly of the Michelson design, which employs a beam splitter to divide the IR beam between fixed and moving mirrors [21]. The recombined beams create an interference pattern (interferogram) that encodes all spectral information, which is subsequently transformed into a conventional intensity-versus-wavenumber spectrum through a Fast Fourier Transform (FFT) algorithm [21].
The critical advantages of FTIR over previous dispersive IR instruments include three fundamental benefits known as Fellgett's (multiplex) advantage, Jacquinot's (throughput) advantage, and Connes' advantage [21]. Fellgett's advantage refers to the signal-to-noise ratio improvement gained by measuring all wavelengths simultaneously rather than sequentially. Jacquinot's advantage results from higher energy throughput due to the absence of narrow slits required in dispersive instruments. Connes' advantage provides exceptional wavelength accuracy and reproducibility through the use of an internal laser reference for interferometer calibration [21] [22]. These combined advantages yield spectra with higher signal-to-noise ratios, better resolution, faster acquisition times, and more reliable calibration transfer compared to dispersive technologies, making FTIR particularly suited for detecting trace-level organic pollutants across diverse sample types [21].
Key Instrumentation Components
Modern FTIR instruments share fundamental components regardless of specific configuration: an infrared radiation source, interferometer, sample compartment, detector, and computer system for Fourier transformation. The IR source typically consists of an inert solid heated electrically to promote thermal emission, providing broadband infrared radiation [22]. The interferometer, most commonly a Michelson design with a beam splitter and mirror arrangement, creates the interference patterns essential to the technique. Various detectors, including deuterated triglycine sulfate (DTGS) and mercury cadmium telluride (MCT) detectors, convert the infrared signal into electrical signals for digitization [21]. The computer system performs the complex Fourier transform mathematics, converting the time-domain interferogram into a frequency-domain spectrum that scientists can interpret.
For specialized applications in organic pollutant research, FTIR systems can be configured with complementary accessories and technologies. These include hyphenated systems such as TGA-FTIR (thermogravimetric analysis) for studying thermal degradation products [23], FTIR microscopes (micro-FTIR) for analyzing microscopic contaminants like microplastics [21] [24], and portable field instruments for on-site environmental monitoring [24]. These technological advancements have significantly expanded FTIR's applicability across the entire range of environmental sampling scenarios, from controlled laboratory analysis to field-based screening and monitoring campaigns.
Sampling Techniques for Different Physical States
Solid Sample Analysis
Solid samples represent some of the most common matrices analyzed for organic pollutants, including soil sediments, microplastics, polymer fragments, and biological tissues. FTIR offers multiple approaches for solid sample analysis, each with specific advantages for different sample types. Attenuated Total Reflectance (ATR) has emerged as the most popular modern technique for solids, requiring minimal sample preparation [21]. In ATR-FTIR, the sample is pressed against a high-refractive-index crystal (diamond, ZnSe, or Ge), and the IR beam undergoes total internal reflection, creating an evanescent wave that penetrates 1-2 μm into the sample [21]. This shallow penetration depth makes ATR particularly suitable for surface analysis and strongly absorbing materials. Diffuse Reflectance (DRIFTS) is ideal for powdered samples such as soils, catalysts, or environmental particulates, where scattered radiation from rough surfaces is collected [21]. For microplastic identification and other microscopic analyses, micro-FTIR (μ-FTIR) enables high-resolution spatial mapping of heterogeneous solid samples, combining microscopy with spectroscopy to identify and characterize small contaminant particles [21] [24].
Traditional transmission methods remain valuable for solid samples that can be prepared as thin films or KBr pellets. In transmission FTIR, the infrared beam passes directly through a thinly prepared sample, providing high-quality spectra but requiring careful thickness control to avoid total absorption [21]. For specialized applications, specular reflection and grazing angle techniques such as Specular Apertured Grazing Angle (SAGA) IR are employed for analyzing thin films or monolayers on reflective surfaces, enhancing sensitivity for surface contaminants [24]. Photoacoustic FTIR (FT-IR-PAS) offers unique capabilities for analyzing highly absorbing, opaque, or heterogeneous solid materials that challenge conventional reflection or transmission methods [21].
Liquid Sample Analysis
Liquid sampling encompasses environmental waters, organic extracts, oils, and chemical solutions containing dissolved pollutants. Transmission cells with precisely spaced infrared-transparent windows (KBr, NaCl, or BaF₂) represent the traditional approach, where spectrum quality depends on selecting appropriate pathlengths (typically 0.01-1 mm) to avoid total absorption while maintaining adequate signal [21]. ATR-FTIR has largely superseded transmission methods for routine liquid analysis due to significantly simplified operation—liquids need only be placed on the crystal surface without pathlength calibration [21]. The evanescent wave penetration provides natural thickness limitation, making ATR ideal for analyzing aqueous solutions and organic solvents, including identifying petroleum hydrocarbons in water, monitoring chemical reactions, or quantifying pollutants in industrial effluents.
For specialized applications, horizontal ATR accessories enable analysis of volatile liquids without evaporation issues, while flow-through cells facilitate online monitoring of liquid streams in industrial or environmental settings. When analyzing liquids, particularly those containing water, researchers must consider solvent compatibility with accessory materials and account for strong solvent absorptions that may obscure regions of interest. Advanced approaches such as differential spectroscopy can subtract solvent contributions, revealing solute spectra even in complex matrices [21]. In pollutant research, this enables detection of trace organic contaminants in environmental waters despite dominant water absorptions.
Gas Sample Analysis
Gas phase analysis represents one of the most historically significant applications of FTIR spectroscopy, particularly for atmospheric monitoring and industrial emission control. Gas cells with extended pathlengths (typically 2-20 meters) employing White cell or multiple reflection designs enhance sensitivity for trace gas detection by increasing the interaction volume between IR radiation and analyte molecules [21]. These long-path cells use precisely aligned mirrors to fold the beam through the sample multiple times, effectively creating pathlengths of 10 meters or more within compact laboratory instruments [21]. Open-path FTIR configurations extend this concept to field applications, using the atmosphere itself as the sample cell and employing natural IR sources (the sun) or artificial sources for remote sensing of atmospheric pollutants over distances up to several hundred meters [21].
The quantitative analysis of gases benefits from their well-defined rotational-vibrational spectra, featuring sharp absorption lines with minimal matrix effects compared to condensed phases. However, accurate quantification requires careful calibration accounting for pressure broadening and temperature effects [21]. For complex gas mixtures, spectral separation algorithms and multicomponent analysis techniques deconvolute overlapping absorptions, enabling simultaneous quantification of multiple pollutants [23]. Applications range from workplace air monitoring to stack emission measurements and climate research, targeting volatile organic compounds (VOCs), greenhouse gases, toxic industrial chemicals, and combustion products.
Table 1: FTIR Sampling Techniques for Different Physical States
| Sample State | Sampling Technique | Typical Applications | Key Advantages | Limitations |
|---|---|---|---|---|
| Solid | Attenuated Total Reflectance (ATR) | Polymers, soils, powders, biological tissues | Minimal sample preparation; surface-sensitive | Limited penetration depth (~1-2 µm) |
| Diffuse Reflectance (DRIFTS) | Powders, catalysts, soil samples | Minimal preparation; ideal for scattering samples | Particle size effects; quantitative challenges | |
| Transmission | KBr pellets, thin films | High-quality spectra; traditional approach | Labor-intensive preparation; thickness critical | |
| Microspectroscopy (μ-FTIR) | Microplastics, single particles, heterogeneity | Spatial resolution; mapping capability | Small analysis area; longer collection times | |
| Liquid | ATR | Aqueous solutions, oils, organic solvents | No pathlength calibration; easy cleaning | Solvent compatibility with crystal |
| Transmission | Solvent analyses, quantitative work | Established methods; high sensitivity | Fixed pathlength cells; air bubbles problematic | |
| Gas | Long-path Gas Cell | Air monitoring, volatile organics | High sensitivity for trace gases | Pressure and temperature effects |
| Open-path | Atmospheric studies, fence-line monitoring | Real-time field measurements; no sampling | Weather dependent; complex calibration |
Experimental Protocols for Organic Pollutant Analysis
Solid Sample Protocol: Microplastic Analysis in Environmental Matrices
The detection and identification of microplastics in environmental samples represents a growing application of FTIR spectroscopy in pollution research. The analytical workflow begins with sample collection and preparation. Environmental matrices (water, sediment, or biological samples) are first processed to separate microplastic particles through density separation, filtration, and digestion of organic matter [21]. The prepared particles are transferred onto appropriate substrates (e.g., aluminum filters, IR-transparent windows) for analysis. For single-particle identification, micro-FTIR in transmission or reflection mode is employed, with standardized methodologies recommending diffuse reflection for superior accuracy with small particles [21].
Spectral collection parameters must be optimized for microanalysis. A typical protocol uses 4 cm⁻¹ resolution with 32-64 scans to ensure adequate signal-to-noise while maintaining reasonable collection times [21]. Background spectra should be collected from a clean area of the substrate immediately before sample analysis to correct for atmospheric interferences. Following data acquisition, spectra are processed with baseline correction and normalization before comparison to spectral libraries of common polymers (polyethylene, polypropylene, polystyrene, etc.). For comprehensive analysis, automated mapping techniques can characterize heterogeneous samples, collecting thousands of spectra across a defined area to create chemical images showing particle distribution and composition [21]. Quality control measures include analyzing known standards alongside samples and verifying automated identifications through manual spectral inspection.
Liquid Sample Protocol: Hydrocarbon Contamination in Water
The identification and quantification of hydrocarbon pollutants in aqueous environments demonstrates FTIR's capability for liquid sample analysis. The experimental approach begins with sample collection and preservation, avoiding plastic containers that may leach interfering compounds. For direct analysis of oils and non-polar hydrocarbons, ATR-FTIR provides the simplest protocol: a small aliquot (typically 50-100 µL) is placed directly on the ATR crystal, and spectra are collected with 4 cm⁻¹ resolution and 16-32 scans [21]. For trace-level contaminants, liquid-liquid extraction with a compatible solvent (e.g., hexane, dichloromethane) concentrates analytes before analysis.
When analyzing aqueous samples, differential spectroscopy techniques effectively remove strong water absorptions that would otherwise obscure pollutant signals [21]. This requires careful collection of a background spectrum using purified water reference matched to the sample matrix. Quantitative analysis employs the Beer-Lambert law, establishing calibration curves from standard solutions of target pollutants across relevant concentration ranges. For complex hydrocarbon mixtures like petroleum products, spectral band ratios and pattern recognition algorithms can identify specific fuel types and weathering states [25]. Method validation should include determination of detection limits, precision, and accuracy using spiked samples and quality control standards analyzed alongside environmental samples.
Gas Sample Protocol: Volatile Organic Compound (VOC) Monitoring
Gas phase FTIR analysis provides a powerful approach for detecting and quantifying volatile organic pollutants in air. The analytical protocol begins with proper sampling, using either integrated sampling into evacuated gas cells or continuous extraction for real-time monitoring. For laboratory analysis, the evacuated gas cell is filled with the sample to a defined pressure, typically 1-50 torr depending on target concentrations and pathlength [21]. Spectral collection employs higher resolution (1-2 cm⁻¹) than condensed phase analyses to resolve sharp rotational-vibrational bands characteristic of gases [21]. Extended scan times (64-128 scans) improve signal-to-noise for trace-level detection.
Quantitative analysis requires careful attention to pressure and temperature effects, which significantly influence absorption band shapes and intensities [21]. Multicomponent analysis algorithms are essential for complex mixtures where spectral bands frequently overlap [23]. These algorithms employ classical least squares or partial least squares regression to deconvolute contributions from multiple analytes, provided appropriate reference spectra are available [23]. For field applications, open-path systems require additional calibration steps to account for atmospheric conditions and pathlength variations. Method validation should include determination of measurement precision, detection limits for target compounds, and interference testing for common atmospheric constituents like water vapor and carbon dioxide [21].
Data Interpretation and Functional Group Identification
Spectral Interpretation Fundamentals
Interpreting FTIR spectra begins with understanding the two primary spectral regions: the functional group region (4000-1500 cm⁻¹) and the fingerprint region (1500-400 cm⁻¹) [13]. The functional group region contains characteristic absorptions from stretching vibrations of common functional groups like O-H, N-H, C-H, and C=O bonds, providing initial clues about molecular structure [13]. The fingerprint region contains complex patterns resulting from coupled vibrations that are highly specific to the entire molecular structure, enabling definitive compound identification [13]. Systematic interpretation starts at the high-frequency end of the spectrum, identifying obvious peaks in the functional group region before analyzing the more complex fingerprint region [13].
For organic pollutant identification, several key spectral regions provide critical structural information. The hydrocarbon region (3100-2800 cm⁻¹) reveals aromatic and aliphatic C-H stretches, distinguishing different classes of petroleum products [25]. The carbonyl region (1850-1650 cm⁻¹) identifies oxidation products, with specific frequencies differentiating acids, esters, ketones, and aldehydes [25]. The single-bond region (1500-900 cm⁻¹) contains C-O, C-N, and C-C stretches that help identify specific compound classes, while the aromatic region (900-650 cm⁻¹) provides substitution pattern information through out-of-plane C-H bending vibrations [25]. Modern interpretation relies heavily on spectral library searching, where unknown spectra are compared against extensive databases of known compounds, with match quality indicating identification confidence [23] [13].
Characteristic Organic Pollutant Signatures
Different classes of organic pollutants exhibit distinctive FTIR spectral patterns that facilitate their identification in complex environmental samples. Petroleum hydrocarbons show dominant aliphatic C-H stretches between 3000-2800 cm⁻¹, with the ratio of asymmetric to symmetric CH₂ and CH₃ stretches indicating chain length and branching [25]. Weathered petroleum products display additional carbonyl absorptions around 1700 cm⁻¹ from oxidation products. Chlorinated solvents and pesticides exhibit characteristic C-Cl stretches between 800-600 cm⁻¹, with specific patterns indicating different chlorination patterns [25]. Organophosphorus compounds show strong P=O stretches around 1250 cm⁻¹ and P-O-C stretches between 1050-1000 cm⁻¹ [25].
Table 2: Characteristic FTIR Absorptions for Organic Pollutant Functional Groups
| Functional Group | Peak Position (cm⁻¹) | Peak Characteristics | Representative Pollutants |
|---|---|---|---|
| O-H stretch | 3200-3550 (H-bonded) | Strong, broad | Alcohols, phenols, carboxylic acids |
| C-H stretch (aromatic) | 3050-3100 | Medium to weak | Aromatic hydrocarbons, PAHs |
| C-H stretch (aliphatic) | 2840-3000 | Medium | Petroleum hydrocarbons, alkanes |
| C≡N stretch | 2222-2260 | Weak to medium, sharp | Nitriles, cyanide compounds |
| C=O stretch | 1690-1760 | Strong, sharp | Aldehydes, ketones, carboxylic acids |
| C=C stretch (aromatic) | 1500-1600 | Variable | Aromatic compounds, PAHs |
| N-O stretch | 1500-1550 | Strong | Nitro compounds, explosives |
| C-O stretch | 1020-1250 | Strong, sharp | Alcohols, esters, ethers |
| C-Cl stretch | 700-800 | Strong, sharp | Chlorinated solvents, pesticides |
| C-Br stretch | 515-690 | Strong | Brominated flame retardants |
Polycyclic aromatic hydrocarbons (PAHs) display a combination of aromatic C-H stretches above 3000 cm⁻¹ and aromatic C=C stretches between 1600-1450 cm⁻¹, with specific patterns indicating ring condensation. Polymer contaminants including microplastics exhibit characteristic patterns throughout the spectrum, with polyethylene showing dominant methylene peaks, polystyrene displaying aromatic C-H stretches, and polyvinyl chloride exhibiting C-Cl stretches [21] [24]. For complex mixtures, spectral subtraction techniques isolate individual component spectra, while multivariate statistical methods like principal component analysis (PCA) classify samples based on their overall spectral patterns [23].
Advanced Applications and Research Innovations
Novel Research Applications
FTIR spectroscopy continues to evolve with technological advancements, enabling new applications in organic pollutant research. In biomedical contexts, researchers are developing universal IR biomarkers for disease detection, with recent studies identifying specific band ratios (particularly 1740/1236 cm⁻¹) that effectively differentiate cancerous and non-cancerous tissues in colorectal cancer [26]. This approach demonstrates how FTIR can detect molecular-level biochemical changes induced by environmental pollutant exposure. In food safety, FTIR has emerged as a cost-effective tool for toxic metal profiling by identifying metal-induced alterations in biomolecular structures, though it requires complementary techniques for direct metal quantification [8].
Advanced imaging techniques push spatial resolution to the micrometer scale, enabling microplastic identification and characterization in environmental samples [21] [24]. Synchrotron-based FTIR microspectroscopy provides exceptional signal-to-noise at diffraction-limited spatial resolutions, allowing researchers to map pollutant distribution within individual cells or soil microaggregates [21]. For industrial applications, operando FTIR methods monitor catalytic degradation of organic pollutants in real-time, providing insights into reaction mechanisms and intermediate formation [21]. These advanced applications highlight FTIR's expanding role in addressing complex environmental challenges through molecular-level characterization.
Emerging Methodological Innovations
Methodological innovations significantly enhance FTIR's capabilities for organic pollutant research. Portable FTIR systems equipped with battery power and ruggedized designs enable field-based analysis, with instruments like the MOBILE-IR II providing laboratory-quality spectra in remote locations [24]. These field-deployable systems facilitate rapid screening and decision-making without the delays associated with laboratory transport and analysis. Automated analysis protocols improve consistency between operators, particularly for complex mixture analysis where traditional spectral subtraction introduces user-dependent variability [23]. Random mixture algorithms adapted from X-ray imaging techniques automatically identify components and calculate area percentages without subjective interpretation [23].
The integration of FTIR with complementary analytical techniques creates powerful hyphenated systems for comprehensive characterization. TGA-FTIR combines thermal decomposition with real-time gas analysis, identifying volatile products from polymer degradation or soil organic matter combustion [23]. GC-FTIR provides separation power alongside molecular specificity, resolving complex mixtures before individual component identification. Chemometric advancements, including machine learning algorithms like Elastic Net regularization, identify the most diagnostically significant spectral features from large datasets, improving classification accuracy for complex environmental samples [26]. These methodological innovations collectively expand FTIR's utility across the entire spectrum of organic pollutant research, from fundamental investigations to applied environmental monitoring.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Materials for FTIR Analysis of Organic Pollutants
| Item | Function | Application Notes |
|---|---|---|
| ATR Crystals (Diamond, ZnSe, Ge) | Enables attenuated total reflectance measurements | Diamond: universal use; ZnSe: avoids protein absorption; Ge: high refractive index for hard materials |
| IR-Transparent Windows (KBr, NaCl, BaF₂) | Construction of liquid and gas cells | KBr: broad range but hygroscopic; NaCl: economical; BaF₂: resistant to water, limited range |
| Solid Sample Accessories (DRIFTS cups, pellet dies) | Hold powdered samples for analysis | DRIFTS: minimal preparation; Pellet dies: create KBr disks for transmission |
| Gas Cells (Various pathlengths) | Contain gas samples for analysis | Short path (10 cm): high concentrations; Long path (10 m): trace gas detection |
| Spectral Libraries | Reference for compound identification | Commercial and custom libraries for pollutants, polymers, and environmental contaminants |
| Chemometrics Software | Multivariate data analysis | PCA, PLS, clustering algorithms for complex mixture analysis |
Visualizing FTIR Workflows
The following diagram illustrates the generalized FTIR experimental workflow for organic pollutant analysis across different sample states, highlighting the parallel pathways for solids, liquids, and gases:
FTIR Analysis Workflow for Organic Pollutants
The strategic selection of sampling technique based on physical state ensures optimal data quality while minimizing analytical artifacts. For heterogeneous solid mixtures, additional spatial separation through microscopy or spectral separation via multicomponent algorithms may be incorporated before final interpretation [23]. The workflow emphasizes proper background referencing specific to each sampling approach, as atmospheric corrections (particularly for water vapor and CO₂) are essential for accurate library matching and quantification [21].
From Theory to Practice: FTIR Methodologies for Real-World Pollutant Analysis
The accurate identification of organic pollutants in environmental samples using Fourier Transform Infrared (FTIR) spectroscopy is a cornerstone of modern analytical chemistry. However, the reliability of this identification is profoundly dependent on the sample preparation techniques employed prior to analysis. Proper sample preparation is crucial for obtaining high-quality spectra that accurately represent the sample's chemical composition without artifacts introduced by the preparation method itself. Within the context of a broader thesis on how FTIR identifies organic pollutants, this technical guide examines three fundamental preparation approaches: filtration, Attenuated Total Reflectance (ATR), and transmission methods. These techniques enable researchers to effectively analyze diverse environmental matrices, from wastewater and soil to biological tissues, for the presence of contaminants such as microplastics, persistent organic pollutants (POPs), and hydrocarbon-based compounds [27] [28].
The selection of an appropriate preparation method directly influences key analytical outcomes, including detection limits, spectral quality, and the ability to identify specific functional groups characteristic of organic pollutants. As FTIR continues to evolve as a tool for environmental forensics, understanding the nuances of these techniques becomes paramount for researchers, scientists, and drug development professionals who require robust, reproducible methodologies for contamination assessment and regulatory compliance [8] [29].
Core FTIR Sample Preparation Techniques
Filtration Methods for Particulate Pre-concentration
Filtration serves as a critical pre-concentration step in FTIR analysis, particularly for heterogeneous environmental samples containing suspended particulate matter, such as microplastics in water samples. This process enables the isolation and concentration of target analytes from a liquid matrix onto a filter substrate compatible with subsequent FTIR analysis. The filtration method is especially valuable when analyzing samples with low pollutant concentrations, as it increases the probability of detection by concentrating the analytes into a smaller analysis area [27] [4].
In practice, water samples are vacuum-filtered through membranes with pore sizes selected based on the target particle size range. For microplastic analysis, studies often use filters with pore sizes ranging from 0.45 to 20 micrometers, depending on the target size fraction. After filtration, the retained particles can be transferred to FTIR-compatible substrates or analyzed directly on specialized filter materials that minimize spectral interference [27]. For complex matrices with high organic content, such as wastewater or sludge, filtration is often preceded by digestion steps using reagents like Fenton's reagent to eliminate interfering biological material that could obscure the spectral signatures of target pollutants [27].
Attenuated Total Reflectance (ATR) Technique
The ATR technique has gained significant prominence in FTIR analysis of organic pollutants due to its minimal sample preparation requirements and versatility across sample types. This method operates on the principle of total internal reflection, where an infrared beam passes through an Internal Reflection Element (IRE) crystal with a high refractive index (e.g., diamond, zinc selenide, or germanium) [30]. The beam interacts with the sample in contact with the crystal surface, penetrating only 1-2 micrometers into the sample, which eliminates issues related to sample thickness that plague transmission methods [30] [31].
For solid environmental samples such as soil, microplastic particles, or contaminated biological tissues, preparation involves direct placement on the ATR crystal with application of consistent pressure to ensure optimal contact. Liquid samples, including water extracts or solvent leachates from environmental samples, are applied directly as droplets onto the crystal surface [32] [30]. The minimal preparation makes ATR particularly advantageous for analyzing complex environmental samples where maintaining the sample's original state is crucial for accurate contamination assessment. Recent advances in ATR-FTIR imaging have further enhanced its application, enabling the mapping of pollutant distribution on heterogeneous environmental samples with high spatial resolution [31].
Transmission Technique
Transmission FTIR represents the classical approach to infrared spectroscopy and remains widely used for specific applications in organic pollutant analysis. In this method, infrared light passes directly through a prepared sample, and the transmitted light is measured to generate the absorption spectrum [30]. The fundamental requirement for transmission FTIR is that samples must be sufficiently thin (typically <20 micrometers for solids) to prevent complete absorption of the infrared beam, which necessitates more extensive preparation compared to ATR [31].
For solid environmental samples, the KBr pellet method is commonly employed, where 1-2 mg of the sample is finely ground and mixed with 100-200 mg of potassium bromide (KBr), then pressed under high pressure to form a transparent pellet [32]. Alternatively, solid samples can be prepared as thin films by dissolving them in a suitable solvent and casting onto IR-transparent windows. Liquid samples are analyzed using sealed liquid cells with path lengths optimized to avoid saturation of strong absorption bands, with window materials (e.g., NaCl, KBr, or CaF₂) selected based on the sample's solubility and spectral requirements [32] [30]. Gas analysis employs specialized gas cells with long path lengths to enhance sensitivity for detecting volatile organic compounds [32].
Comparative Analysis of Preparation Methods
The selection of an appropriate FTIR sample preparation method requires careful consideration of multiple factors, including sample type, analytical objectives, and available resources. The table below provides a systematic comparison of the three core techniques:
Table 1: Comprehensive Comparison of FTIR Sample Preparation Methods
| Parameter | Filtration | ATR | Transmission |
|---|---|---|---|
| Primary Application | Pre-concentration of particulates from liquids [27] | Direct analysis of solids, liquids, pastes [32] | High-quality spectral analysis of prepared samples [30] |
| Sample Preparation Complexity | Moderate (requires filtration apparatus) | Minimal (direct application) [30] | High (grinding, pressing, or cell assembly) [30] |
| Typical Sample Thickness | Filter-dependent | ~1-2 μm penetration depth [30] | <20 μm for solids [31] |
| Analysis Time | Moderate to High (including filtration time) | Low (minimal preparation) [29] | High (extensive preparation required) [30] |
| Spectral Reproducibility | Variable (depends on filter homogeneity) | High [30] | Variable (depends on preparation skill) [30] |
| Spectral Quality | Good (potential for filter background) | Excellent (modern instruments) [31] | Excellent (traditional gold standard) [30] |
| Sample Recovery | Difficult (embedded in filter) | Easy (non-destructive) [30] | Difficult (destructive for KBr pellets) |
| Spectral Libraries | Custom libraries often needed | Growing availability [30] | Extensive historical libraries [30] |
| Ideal for Aqueous Samples | Yes (pre-concentration) | Limited (water strong IR absorber) | Challenging (requires short path lengths) [31] |
Each method presents distinct advantages and limitations that determine its suitability for specific research scenarios. ATR-FTIR offers significant operational efficiencies with minimal sample preparation, making it ideal for rapid screening of diverse sample types and for analyzing materials that are difficult to prepare using traditional transmission methods [30]. The non-destructive nature of ATR also allows for sample recovery for subsequent analyses, an important consideration when dealing with limited or evidentiary samples. Furthermore, ATR enables the analysis of highly absorbing, opaque samples that would be unsuitable for transmission measurements [31].
In contrast, transmission FTIR, despite its more labor-intensive preparation requirements, remains the method of choice when library matching against extensive historical databases is essential for compound identification [30]. The KBr pellet method specifically provides excellent spectral quality for homogeneous solid samples and enables precise control over sample concentration in the analysis path. However, the hygroscopic nature of KBr requires careful handling to avoid moisture absorption, which can introduce spectral artifacts [30].
Filtration methods occupy a specialized niche in environmental analysis, serving primarily as a pre-concentration technique that bridges sample collection and subsequent FTIR analysis via either ATR or transmission methods. Their value is particularly evident in the analysis of microplastics in water samples, where concentrating dispersed particles onto a filter surface enables morphological and chemical characterization that would be impossible in dilute solutions [27] [4].
Table 2: Method Selection Guide Based on Sample Type and Research Objective
| Sample Type | Primary Research Objective | Recommended Method | Key Considerations |
|---|---|---|---|
| Wastewater/Environmental Water | Microplastic quantification | Filtration + ATR [27] | Combine with organic digestion for complex matrices |
| Soil/Sediment | Organic pollutant screening | ATR [4] | Minimal preparation; direct analysis possible |
| Soil/Sediment | High-resolution polymer identification | Transmission (KBr pellet) [4] | Provides superior spectral quality for mixed polymers |
| Biological Tissues | Contamination mapping | ATR-FTIR imaging [31] | Preserves spatial distribution of contaminants |
| Liquid Extracts/Leachates | Organic pollutant profiling | ATR [28] | Avoids cell assembly issues; small volume required |
| Air Particulates | Source identification | Transmission (KBr pellet) | Enhanced spectral library matching |
| Complex Polymer Mixtures | Degradation product identification | Transmission (KBr pellet) | Superior resolution of overlapping peaks |
Experimental Protocols for Organic Pollutant Analysis
Protocol 1: Microplastic Analysis in Water Samples Using Filtration and ATR-FTIR
This protocol details a method for identifying and quantifying microplastic particles in water samples, combining filtration for pre-concentration and ATR-FTIR for chemical identification [27] [4].
Materials and Reagents:
- Glass filtration apparatus with vacuum source
- IR-transparent filter membranes (e.g., aluminum oxide, polycarbonate)
- Fenton's reagent (for organic-rich samples): 30% H₂O₂ and FeSO₄ catalyst [27]
- Zinc chloride (ZnCl₂) for density separation (optional)
- ATR-FTIR spectrometer with diamond crystal
- Deionized water for rinsing
Procedure:
- Sample Digestion (if required): For samples with high organic content, add Fenton's reagent (30% H₂O₂ with FeSO₄ catalyst) to digest biological material. Perform multi-digestion treatments for complex matrices [27].
- Density Separation (optional): Add zinc chloride (ZnCl₂) solution to separate microplastics from mineral particles through flotation [27].
- Filtration: Filter the treated water sample through an IR-transparent membrane using a vacuum filtration setup. The pore size should be selected based on the target size fraction (typically 0.45-20 μm).
- Filter Preparation: Carefully transfer the filter to the ATR-FTIR instrument, ensuring the collected particles are in contact with the crystal surface.
- ATR-FTIR Analysis: Apply consistent pressure to ensure good contact between the filter and the ATR crystal. Collect spectra in the range of 4000-600 cm⁻¹ with 4 cm⁻¹ resolution. For heterogeneous samples, perform mapping across multiple points.
- Spectral Interpretation: Identify polymer types by characteristic absorption bands: PP/PS (1637 cm⁻¹), PE (1031 cm⁻¹), PET (582 cm⁻¹), and polyamide (3448 cm⁻¹ for -OH groups) [4].
Protocol 2: Analysis of Organic Pollutants in Soil Using Transmission FTIR
This protocol describes the preparation of soil samples for transmission FTIR analysis, particularly useful for identifying organic pollutants and their interactions with soil components [4] [33].
Materials and Reagents:
- Agate mortar and pestle
- Potassium bromide (KBr), spectroscopic grade
- Hydraulic pellet press
- Vacuum die for pellet formation
- FTIR spectrometer with transmission accessory
Procedure:
- Soil Preparation: Air-dry soil samples and sieve through a 2 mm mesh to remove large debris. Homogenize using an agate mortar and pestle [4].
- Powder Grinding: Grind approximately 1-2 mg of soil sample into a fine powder using the agate mortar and pestle.
- KBr Mixture Preparation: Mix the ground soil sample with 100-200 mg of dry KBr powder (spectroscopic grade) in a 1:100 sample-to-KBr ratio. Grind thoroughly to ensure homogeneous distribution.
- Pellet Formation: Transfer the mixture to a vacuum die and apply pressure of approximately 10-15 tons/cm² using a hydraulic press for 1-2 minutes to form a transparent pellet.
- FTIR Analysis: Place the KBr pellet in the sample holder of the FTIR spectrometer. Collect transmission spectra in the range of 4000-400 cm⁻¹ with 4 cm⁻¹ resolution. Run a pure KBr pellet as background reference.
- Data Interpretation: Analyze characteristic absorption bands for soil organic matter (2920 cm⁻¹ and 2850 cm⁻¹ for aliphatic C-H; 1650 cm⁻¹ for aromatic C=C; 1720 cm⁻¹ for C=O in carboxylic acids) and specific organic pollutants based on their functional groups [33].
Protocol 3: Direct Analysis of Plastic Litter Leachates Using ATR-FTIR
This protocol outlines the procedure for analyzing organic pollutants desorbed from plastic litter using ATR-FTIR, particularly relevant for assessing emerging contaminants in marine environments [28].
Materials and Reagents:
- ATR-FTIR spectrometer with diamond crystal
- Glass containers for leaching experiments
- Artificial seawater or natural seawater samples
- Stir bar sorptive extraction (SBSE) equipment (optional)
- GC-MS system for validation (optional)
Procedure:
- Leachate Preparation: Place plastic litter samples in glass containers with artificial or natural seawater at a defined liquid-to-solid ratio. Agitate for 24-48 hours under controlled temperature [28].
- Sample Concentration (optional): For low-concentration analytes, employ stir bar sorptive extraction (SBSE) using a polydimethylsiloxane (PDMS)-coated magnetic stir bar to concentrate hydrophobic organic pollutants.
- ATR-FTIR Analysis: Place a droplet of the leachate directly onto the ATR crystal. For SBSE extracts, elute the concentrated analytes with a small volume of organic solvent before deposition.
- Spectral Acquisition: Collect ATR-FTIR spectra with 4 cm⁻¹ resolution over 32 scans. Ensure the ATR crystal is thoroughly cleaned between samples.
- Contaminant Identification: Identify characteristic peaks of organic pollutants: fragrances (OTNE: 1270, 1170 cm⁻¹), UV filters (1270, 1170 cm⁻¹), PAHs (1600, 1500 cm⁻¹ for aromatic rings) [28].
- Validation (optional): Confirm identifications using GC-MS analysis of parallel samples for targeted quantification of specific organic pollutants.
Workflow Visualization
Figure 1: FTIR Sample Preparation Workflow for Organic Pollutant Analysis
Research Reagent Solutions
Table 3: Essential Reagents and Materials for FTIR Sample Preparation
| Reagent/Material | Function | Application Examples |
|---|---|---|
| Potassium Bromide (KBr) | IR-transparent matrix for pellet preparation | Transmission analysis of solid samples [32] |
| Fenton's Reagent (H₂O₂ + FeSO₄) | Digestion of organic matter in environmental samples | Wastewater, sludge, and biota-rich samples [27] |
| Zinc Chloride (ZnCl₂) | Density separation medium for microplastic isolation | Separation of polymers from mineral particles [27] |
| ATR Crystals (Diamond, ZnSe, Ge) | Internal Reflection Element for ATR-FTIR | Direct analysis of solids, liquids, and complex samples [30] |
| IR-transparent Windows (NaCl, KBr, CaF₂) | Containment of samples in transmission cells | Liquid and gas analysis [32] |
| Polydimethylsiloxane (PDMS) | Extraction phase for stir bar sorptive extraction | Pre-concentration of hydrophobic organic pollutants [28] |
The strategic selection and proper implementation of sample preparation techniques—filtration, ATR, and transmission methods—are fundamental to the successful application of FTIR spectroscopy in organic pollutant research. Each method offers distinct advantages that cater to specific sample types and analytical requirements. Filtration enables the pre-concentration of particulate pollutants from aqueous matrices, ATR provides rapid analysis with minimal sample preparation across diverse sample types, and transmission methods deliver high-quality spectra compatible with extensive reference libraries. Understanding the principles, applications, and limitations of these techniques empowers researchers to design robust analytical workflows that generate reliable data for environmental monitoring, contamination assessment, and regulatory decision-making. As FTIR technology continues to evolve, particularly with advancements in ATR imaging and computational spectroscopy, these sample preparation methods will remain essential components in the analytical toolkit for identifying and characterizing organic pollutants across environmental compartments.
Microplastic pollution has emerged as a critical environmental challenge, with the persistence and transformation of these particles in natural ecosystems being a primary focus of scientific inquiry. The durability of plastic materials, while beneficial for applications, becomes environmentally detrimental when plastic waste fragments into microplastics (MPs) that accumulate in terrestrial, freshwater, and marine environments [3] [34]. Fourier Transform Infrared (FTIR) spectroscopy has established itself as an indispensable analytical technique for tracking microplastic weathering and degradation processes, providing molecular-level insights into the chemical transformations that occur as plastics environmental exposure [3] [21]. This case study examines the specialized application of FTIR spectroscopy for investigating microplastic aging mechanisms, with particular emphasis on quantitative assessment approaches, experimental methodologies, and data interpretation frameworks that enable researchers to decipher the complex lifecycle of plastic pollutants in the environment.
The significance of this research domain stems from the profound influence of weathering on microplastic behavior and impact. Weathering significantly alters the physicochemical properties of MPs, critically affecting their sorption behavior toward pollutants and increasing the potential for ingestion by organisms, including humans [3]. These transformations ultimately determine the environmental fate and ecological risks associated with plastic pollution. Within this context, FTIR spectroscopy serves as a powerful tool that enables researchers to not only identify polymer composition but also to quantitatively track the progression of degradation through established spectroscopic indicators [3] [21]. This case study provides a comprehensive technical examination of FTIR-based approaches for microplastic weathering analysis, with specific attention to experimental protocols, data interpretation methods, and practical applications within environmental research.
FTIR Fundamentals for Microplastic Analysis
Fourier Transform Infrared (FTIR) spectroscopy operates on the principle of measuring molecular vibrations through infrared light absorption, generating characteristic spectral fingerprints that enable material identification and characterization [21] [35]. When IR radiation interacts with a sample, specific frequencies are absorbed corresponding to molecular bond vibrations, including stretching, bending, or twisting of dipoles [21]. In modern FTIR instruments, a broadband IR source passes through a Michelson interferometer containing a moving mirror that produces an interferogram—a complex pattern of constructive and destructive interference that encodes all spectral frequencies simultaneously. This interferogram undergoes mathematical transformation via a fast Fourier transform (FFT) algorithm to produce an intensity-versus-wavenumber spectrum [21].
For microplastic analysis, FTIR spectroscopy offers several distinct advantages, including non-destructive analysis, minimal sample preparation, and the ability to provide both qualitative identification and quantitative assessment of weathering extent [3] [21]. The technique excels particularly in detecting the formation of oxygen-containing functional groups that result from photo-oxidation and other degradation processes, serving as direct indicators of polymer aging [3]. Different sampling geometries can be employed based on sample characteristics and analytical requirements:
- Attenuated Total Reflectance (ATR): The most popular modern technique for solid samples, requiring minimal preparation and providing information from sample surfaces [3] [21]
- Transmission: Suitable for thin films or particles suspended on filters, though more extensive sample preparation may be required [21]
- Diffuse Reflectance (DRIFTS): Particularly effective for powdered samples or rough surfaces [21]
- Microspectroscopy (μ-FT-IR): Essential for analyzing individual microplastic particles, enabling correlation of morphological and chemical information [21]
The resulting infrared spectra serve as molecular fingerprints, with specific absorption bands corresponding to characteristic functional groups within polymer structures. Careful analysis of spectral changes enables researchers to track degradation processes through the appearance, disappearance, or modification of these absorption bands [3] [34].
Weathering Indicators and Degradation Mechanisms
Chemical Transformations in Weathered Microplastics
Environmental weathering induces fundamental chemical changes in plastic polymers, primarily through photo-oxidation driven by ultraviolet radiation, thermal degradation, and biological processes [3]. These mechanisms progressively alter polymer chemistry by breaking molecular bonds and introducing oxygen-containing functional groups. The formation of carbonyl (C=O) groups represents the most significant indicator of polyolefin photo-oxidation, as UV radiation initiates free radical reactions that ultimately produce ketones, aldehydes, and carboxylic acids along the polymer backbone [3]. Simultaneously, hydroxyl (O-H) groups form through secondary oxidation processes or water adsorption into more hydrophilic aged surfaces. The carbon-oxygen (C-O) bonds associated with alcohols, esters, and ethers also increase as fragmentation progresses [3].
The following diagram illustrates the primary chemical pathways in microplastic weathering:
Quantitative FTIR Assessment of Weathering
FTIR spectroscopy enables quantitative assessment of microplastic weathering through established spectral indices that track the progression of chemical changes. These indices are calculated from specific absorbance band ratios that correspond to the formation of oxidative functional groups [3]:
Table 1: FTIR Spectral Indices for Microplastic Weathering Assessment
| Index Name | Formula | Spectral Regions | Interpretation | Application Examples |
|---|---|---|---|---|
| Carbonyl Index (CI) | Absorbance at ~1710 cm⁻¹ / Reference peak | Carbonyl: 1690-1810 cm⁻¹Reference: Polymer-specific | Increases with UV exposure; indicates oxidation extent | PE pellets: CI 0.05-0.26 in freshwater [3] |
| Hydroxyl Index (HI) | Absorbance at ~3400 cm⁻¹ / Reference peak | Hydroxyl: 3100-3700 cm⁻¹Reference: Polymer-specific | Reflects hydroxyl group formation; increases with weathering | PE fragments: HI 5.90±2.57; Lines: HI 11.51 [3] |
| Carbon-Oxygen Index (COI) | Absorbance at ~1100-1300 cm⁻¹ / Reference peak | C-O: 1100-1300 cm⁻¹Reference: Polymer-specific | Indicates C-O bond formation in alcohols, esters | PS fragments: COI increases with irradiation [3] |
The reference peaks used for normalization are polymer-specific, typically employing intrinsic vibrational modes that remain stable throughout degradation, such as the C-H stretching bands between 2800-3000 cm⁻¹ for polyolefins [3]. The sensitivity of these indices to different morphological forms is well-documented, with pellets typically showing higher CI values compared to fragments, while fragments exhibit greater HI and COI values [3]. This differential susceptibility highlights the complex relationship between polymer morphology, surface area, and degradation pathways.
Experimental Protocols for FTIR-Based Weathering Studies
Microplastic Sampling and Preparation
Standardized sampling and preparation protocols are essential for obtaining reproducible FTIR spectra that enable accurate weathering assessment. For environmental microplastics, collection typically involves plankton nets for water column sampling or sediment collection systems for benthic studies, followed by density separation, filtration, and visual sorting under microscopy [3] [34]. For controlled aging experiments, pristine polymer materials are prepared through fragmentation or used as manufactured, followed by systematic exposure to environmental stressors [3].
Critical sample preparation steps for FTIR analysis include:
- Cleaning: Remove adhering biological material and inorganic contaminants through enzymatic treatment or mild oxidative processes without damaging polymer surfaces
- Size Fractionation: Sieve samples to specific size classes (e.g., 1-5 mm) to account for size-dependent weathering patterns [3]
- Drying: Completely remove water through air-drying or nitrogen flux to eliminate spectral interference from O-H stretching and bending vibrations [36]
- Substrate Selection: Mount particles on IR-transparent substrates (KBr, BaF₂ windows) for transmission mode or directly place on ATR crystal for surface analysis [21]
For ATR-FTIR analysis, which has become the predominant technique for microplastic characterization, ensuring optimal contact between the sample and ATR crystal is essential for obtaining high-quality spectra. Applied pressure must be consistent across samples to avoid spectral artifacts, and crystal background scans should be acquired immediately before sample measurement to minimize atmospheric interference [3] [21].
Controlled Aging Experimental Design
Laboratory-based aging studies provide controlled environments for investigating specific degradation mechanisms and validating FTIR weathering indices. Standard protocols involve exposing pristine microplastics to simulated environmental conditions, with periodic sampling for FTIR analysis [3].
Table 2: Experimental Parameters for Controlled Aging Studies
| Parameter | Standard Conditions | Accelerated Aging | Monitoring Protocol |
|---|---|---|---|
| UV Exposure | UVA lamps (340 nm), 22°C, 60% humidity | Higher irradiance or UVB spectrum | FTIR sampling at 0, 5, 10, 15, 20 days [3] |
| Thermal Aging | Constant 45°C in air-circulated oven | Higher temperatures (60-80°C) | Pre- and post-exposure FTIR analysis [3] |
| Aqueous Aging | Natural or artificial seawater, controlled temperature | Elevated temperature, mechanical agitation | Sampling at regular intervals up to 73+ days [34] |
| Biological Aging | Microbial inoculum from relevant environments | Optimized nutrient conditions | Pre- and post-incubation analysis, enzymatic assays |
A comprehensive experimental workflow integrates these elements to systematically investigate weathering processes:
Controlled experiments typically employ a factorial design that systematically varies exposure intensity, duration, and environmental parameters to develop predictive models of degradation behavior. This approach enables researchers to isolate the effects of specific environmental stressors and establish causal relationships between exposure conditions and chemical transformations [3].
Data Analysis and Interpretation
Spectral Processing and Quality Control
Robust data analysis begins with comprehensive spectral processing to eliminate artifacts and enhance relevant chemical information. Essential preprocessing steps include:
- Atmospheric Compensation: Subtract reference spectra of ambient atmosphere to minimize contributions from atmospheric CO₂ (2350 cm⁻¹) and water vapor (3900-3400 cm⁻¹ and 1900-1300 cm⁻¹) [21]
- Baseline Correction: Apply linear or polynomial functions to eliminate scattering effects and offset drift, particularly crucial for quantitative analysis [36] [21]
- Smoothing: Implement Savitzky-Golay or similar algorithms to improve signal-to-noise ratio without distorting spectral features [36]
- Normalization: Scale spectra to consistent intensity using internal standards (vector normalization) or stable polymer bands to enable comparative analysis [3] [36]
- Spectral Derivatives: Calculate first or second derivatives to resolve overlapping bands and enhance subtle spectral features [36]
Quality control measures should include verification of signal-to-noise ratios (>20:1 for most applications), examination of absolute absorption values (typically 0.1-1.0 AU for optimal quantification), and assessment of band shape consistency across replicate measurements [21]. For ATR measurements, correction for wavelength-dependent penetration depth should be applied, particularly when comparing samples across different sessions or instruments [21].
Weathering Index Calculation and Statistical Analysis
Following spectral preprocessing, weathering indices are calculated according to established formulas (Table 1) and subjected to statistical analysis to identify significant trends and relationships. For environmental samples, index values should be compared against both pristine controls and samples subjected to controlled laboratory aging to establish relative weathering states [3]. Multivariate statistical approaches, including principal component analysis (PCA) and hierarchical clustering, can identify patterns in complex spectral datasets and classify samples according to degradation state [21] [35].
Longitudinal data from time-series experiments should be analyzed using regression models to quantify degradation rates. Research indicates that carbonyl formation in polyethylene under UV exposure often follows pseudo-zero-order kinetics during initial stages, with CI increases of 30% or more observed during accelerated aging [3]. For compostable plastics, isotopic approaches combined with FTIR have estimated complete degradation of starch components within 124-180 days in marine environments [34].
Research Reagent Solutions and Materials
Successful execution of FTIR-based microplastic weathering studies requires specific reagents and materials optimized for spectroscopic analysis and environmental simulation.
Table 3: Essential Research Materials for Microplastic Weathering Studies
| Category | Specific Items | Function/Application | Technical Considerations |
|---|---|---|---|
| Reference Materials | Pristine polymer pellets (PE, PP, PS)Certified reference materials | Positive controls and baseline measurements | Source from accredited manufacturers; characterize before use [3] |
| FTIR Accessories | ATR crystals (diamond, ZnSe)IR-transparent windows (KBr, BaF₂) | Spectral acquisition in various modes | Diamond ATR most durable for environmental samples [21] |
| Sample Preparation | Enzymatic cleaning solutionsDensity separation salts (NaCl, NaI)Anodisc filters | Contaminant removal, MP isolation, Filtration | Enzymatic treatment preserves polymer chemistry [3] |
| Aging Simulation | UVA/UVB lamps (340-315 nm)Artificial seawater formulationsTemperature-controlled incubators | Accelerated photo-aging, Aquatic degradation studies | Match solar spectrum for environmental relevance [3] [34] |
| Calibration Standards | Polystyrene narrow standardsOxidized polyethylene reference | Molecular weight correlation, Oxidation quantification | Establish calibration curves for quantitative work [3] |
FTIR spectroscopy has established itself as a cornerstone analytical technique for tracking microplastic weathering and degradation across environmental compartments. The methodology provides unparalleled capability to monitor chemical transformations non-destructively, quantitatively, and with high molecular specificity. Through standardized protocols for spectral acquisition, index calculation, and experimental design, researchers can generate comparable data across studies and ecosystems, advancing our understanding of plastic persistence globally. The integration of FTIR with complementary techniques, including isotope analysis, computational modeling, and toxicological assessment, will further strengthen its utility in addressing the complex challenges posed by plastic pollution. As environmental regulations evolve and new polymer formulations emerge, FTIR spectroscopy will continue to provide essential analytical capabilities for predicting plastic fate and informing mitigation strategies.
Fourier Transform Infrared (FTIR) spectroscopy has emerged as a powerful analytical technique for identifying molecular structures by detecting their characteristic absorption of infrared radiation [22]. When applied to airborne particulate matter (PM), FTIR spectroscopy, particularly using Attenuated Total Reflectance (ATR) accessories, provides a non-destructive method for characterizing both organic functional groups and inorganic ions in environmental samples [37] [6]. This capability is crucial for understanding the composition, sources, and potential health impacts of air pollution, bridging a critical gap between total mass measurements and molecular-level analysis [38]. The technique's versatility allows researchers to investigate a wide spectrum of atmospheric components, from Saharan dust transported across continents to locally emitted organic pollutants [37]. This case study examines the application of ATR-FTIR spectroscopy for comprehensive PM profiling, focusing on methodological approaches, key findings from field studies, and the technique's integration into broader environmental research initiatives.
Technical Foundation of FTIR Spectroscopy
Fundamental Principles
FTIR spectroscopy operates on the principle that molecules absorb specific frequencies of infrared radiation corresponding to their vibrational energy transitions [22]. When exposed to infrared radiation, sample molecules selectively absorb radiation at characteristic wavelengths that cause changes in their dipole moment, resulting in vibrational energy level transitions from ground to excited states [22]. The resulting absorption spectrum serves as a molecular "fingerprint" that identifies functional groups and chemical bonds present in the sample [39]. The Fourier Transform algorithm converts the raw interferogram signal into an interpretable spectrum, enabling high signal-to-noise ratios and wavenumber accuracy within ±0.01 cm⁻¹ [22].
Modern FTIR spectrometers offer significant advantages over previous generations of infrared instruments, including rapid scan times (approximately 1 second for all frequencies), high resolution (0.1-0.005 cm⁻¹), and a wide scan range (1000-10 cm⁻¹) [22]. The mid-infrared region (4000-400 cm⁻¹) is particularly useful for environmental analysis as it captures the absorption radiation of most organic compounds and inorganic ions found in atmospheric samples [22].
ATR-FTIR Methodology
Attenuated Total Reflectance (ATR) is a sampling technique that enhances FTIR applications for complex environmental samples like PM [37]. In ATR-FTIR, an optically transparent crystal with a high refractive index (the Internal Reflection Element or IRE) enables the IR beam to undergo multiple internal reflections [40]. At each reflection point, an evanescent wave penetrates a short distance (typically 0.5-2 µm) into the sample in contact with the crystal, where it is absorbed by the sample components [40].
The depth of penetration (dₚ) is calculated as: [ dp = \frac{\lambda}{2\pi n1[\sin^2\theta - (n2/n1)^2]^{1/2}} ] where λ is the wavelength, n₁ is the refractive index of the IRE, n₂ is the refractive index of the sample, and θ is the angle of incidence [40].
ATR-FTIR offers several advantages for PM analysis: it requires minimal sample preparation, accommodates small sample sizes, eliminates light scattering issues common with particulate samples, and allows direct analysis of filters without extraction or derivatization [38] [6]. This makes it particularly valuable for analyzing the complex organic fraction of aerosols, where traditional methods like gas chromatography-mass spectrometry (GC-MS) can only identify 10-50% of the organic mass due to analytical limitations [38].
Analytical Approaches for PM Characterization
Spectral Identification of Functional Groups and Ions
ATR-FTIR analysis enables simultaneous identification of inorganic ions and organic functional groups in PM samples through their characteristic vibrational frequencies. The table below summarizes the primary spectral signatures identified in ambient aerosol samples.
Table 1: Characteristic ATR-FTIR Spectral Signatures for PM Components
| Component Type | Specific Compound/Group | Vibrational Frequencies (cm⁻¹) | Assignment |
|---|---|---|---|
| Inorganic Ions | Sulfate (SO₄²⁻) | 603, 615, 670, 1100 | S-O stretching |
| Ammonium (NH₄⁺) | 1414 | N-H bending | |
| Nitrate (NO₃⁻) | 825, 1356 | N-O stretching | |
| Carbonate (CO₃²⁻) | 713, 730, 877 | C-O stretching | |
| Organic Functional Groups | Aliphatic Hydrocarbons | 2850, 2924, 2950 | CH₂, CH₃ stretching |
| Carbonyl Groups | 1690-1810 | C=O stretching | |
| Hydroxyl Groups | 3100-3700 | O-H stretching | |
| Alkenes/Carbon Double Bonds | 1600-1680 | C=C stretching | |
| Clay Minerals | Saharan Dust Components | Specific peaks in fingerprint region | Si-O, Al-O stretching |
Studies of PM samples collected during Saharan dust events have confirmed the presence of clay minerals alongside organic components, demonstrating the technique's capability to characterize both natural and anthropogenic aerosol sources [37]. The "fingerprint region" (1500-500 cm⁻¹) provides particularly distinctive patterns for source identification [3].
Quantitative Assessment Methodologies
Beyond qualitative identification, ATR-FTIR facilitates quantitative analysis of PM components through carefully calibrated approaches. Multivariate chemometric techniques, such as principal components analysis (PCA) and partial least squares (PLS) modeling, enable the quantification of functional groups and inorganic ions in ambient aerosols [14] [6].
The organic mass (OM) to organic carbon (OC) ratio can be determined using FTIR data, providing crucial information about the oxidation state and sources of organic aerosols [38]. This ratio typically ranges from 1.4 to 2.2 for ambient aerosols, with higher values indicating more oxidized organic material [38].
For microplastic analysis, specific degradation indices are calculated to assess the weathering state of environmental particles [3]:
- Carbonyl Index (CI): Ratio of carbonyl peak intensity (~1710 cm⁻¹) to reference peak (~ 1460 cm⁻¹ or 1370 cm⁻¹)
- Hydroxyl Index (HI): Ratio of hydroxyl peak intensity (~3400 cm⁻¹) to reference peak
- Carbon-Oxygen Index (COI): Ratio of C-O stretching intensity (~1000-1300 cm⁻¹) to reference peak
These indices have been successfully applied to track polymer degradation in environmental samples, with studies reporting CI values for polyethylene pellets ranging from 0.05 to 0.26 in freshwater environments [3].
Table 2: Performance Comparison of PM Analytical Techniques
| Technique | Analytical Scope | Detection Limits | Sample Preparation | Key Limitations |
|---|---|---|---|---|
| ATR-FTIR | Organic functional groups, inorganic ions | Varies by component; ~100 ng for some organics | Minimal; direct filter analysis | Limited surface sensitivity (~100 nm film thickness) |
| Ion Chromatography (IC) | Water-soluble ions (SO₄²⁻, NO₃⁻, NH₄⁺) | Low ppb range for most ions | Extraction required | Limited to ionic species only |
| Thermal-Optical Methods (TOM) | Organic Carbon (OC), Elemental Carbon (EC) | ~0.2 µg C/m³ | Filter punching | Does not distinguish organic components |
| GC-MS | Specific organic compounds | ppt-ppb range for targeted compounds | Extensive extraction and derivatization | Identifies only 10-50% of organic mass |
Experimental Protocol for PM Analysis by ATR-FTIR
Sample Collection and Preparation
Proper sample collection and preparation are critical for reliable ATR-FTIR analysis of PM. The following protocol outlines the standard methodology based on published studies:
Sampling Equipment and Materials:
- Low-volume particulate sampler operating at 2.3 m³/h [37]
- Quartz fiber filters (47 mm diameter) for PM collection [37]
- Teflon filters for complementary analysis [38]
- Forceps for filter handling
- Desiccator for filter conditioning (48 hours at 20±1°C and 50±5% RH) [37]
Sample Collection:
- Deploy samplers at strategically selected sites representing different environments (urban, rural, background) [37] [38]
- Conduct 24-hour sampling from midnight to midnight to ensure consistency [37]
- Collect parallel samples on different filter media for complementary analyses (e.g., Teflon for FTIR, quartz for OC/EC) [38]
- Record meteorological parameters (wind speed, direction, temperature) during sampling [37]
Sample Preparation for ATR-FTIR:
- Condition filters for 48 hours in a temperature- and humidity-controlled environment (20±1°C, 50±5% RH) before and after sampling [37]
- Cut filter subsections using clean ceramic scissors for analysis
- Place the filter sample-side down directly on the ATR crystal [37]
- Apply consistent pressure using the ATR pressure tower to ensure good contact [37]
Instrumental Analysis and Data Acquisition
Equipment Specifications:
- FTIR spectrometer with deuterated triglyceride sulfate (DTGS) detector [37]
- ATR accessory with germanium or diamond crystal [3] [37]
- Resolution: 2-4 cm⁻¹ [3] [37]
- Spectral range: 4000-400 cm⁻¹ [37]
- Scans per spectrum: 32 [3] [37]
Data Collection Procedure:
- Background spectrum collection with clean ATR crystal
- Place sample on crystal and ensure proper contact
- Collect sample spectrum with specified parameters (32 scans, 4 cm⁻¹ resolution) [3]
- Repeat for multiple locations on filter to assess homogeneity
- Include quality control samples (field blanks, laboratory blanks) in each batch
Data Processing and Analysis:
- Subtract blank filter spectrum from sample spectrum [38]
- Apply atmospheric correction for CO₂ and water vapor interference
- Identify peak positions and assign to functional groups/ions (Table 1)
- Integrate peak areas for quantitative analysis
- Apply chemometric techniques (PCA, PLS) for complex mixtures [14]
Case Study: PM Analysis in Urban and Rural Environments
Study Design and Sampling Strategy
A comprehensive PM analysis study was conducted in the Pearl River Delta region of South China, representing a typical rapidly developing megacity environment [38]. The investigation employed ATR-FTIR to characterize PM₂.₅ composition at multiple sites during dry seasons to capture pollution episodes. Four strategically selected sampling locations included two urban sites (SZ and YJ) and two rural sites (LM and HX) to enable urban-rural comparisons [38]. Parallel sampling was conducted using both Teflon filters for ATR-FTIR analysis and quartz fiber filters for complementary OC/EC and ion chromatography (IC) analysis, allowing method comparison [38].
The sampling campaign successfully captured a haze event, providing insights into compositional changes during elevated pollution periods. This design enabled researchers to address a key scientific question: whether the fast and cost-effective ATR-FTIR technique could yield comparable results to traditional analyzers (IC and OC/EC) for characterizing PM₂.₅ composition variations, particularly during pollution events [38].
Comparative Performance Assessment
The ATR-FTIR method demonstrated strong capability for screening major PM components when compared with established reference methods. Key findings from the comparative analysis include:
Table 3: ATR-FTIR Performance in PM Component Detection
| PM Component | ATR-FTIR Detection Capability | Correlation with Reference Methods | Limitations/Considerations |
|---|---|---|---|
| Sulfate | Strong detection at 1100 cm⁻¹ | High correlation with IC measurements | Teflon filter interference at 1213-1152 cm⁻¹ |
| Nitrate | Detectable at 825-1356 cm⁻¹ | Good correlation with IC measurements | Semi-volatile nature may lead to losses |
| Ammonium | Identifiable at 1414 cm⁻¹ | Moderate correlation with IC measurements | Overlap with organic peaks possible |
| Organic Aerosols | Aliphatic hydrocarbons at 2850-2950 cm⁻¹ | Good agreement with OC/EC analyzers | Complex mixture requires chemometrics |
| Crustal Material | Carbonates at 713-877 cm⁻¹ | Consistent with dust event observations | May be obscured in heavy pollution |
The study revealed that ATR-FTIR could reliably characterize composition variations in PM₂.₅, particularly during pollution episodes when concentrations were sufficiently high [38]. The technique showed slightly reduced performance at low PM₂.₅ concentrations but remained a valuable screening tool across concentration ranges [38].
The Scientist's Toolkit: Essential Research Reagents and Materials
Successful implementation of ATR-FTIR for PM analysis requires specific materials and reagents optimized for environmental sampling and spectroscopic characterization.
Table 4: Essential Research Reagents and Materials for ATR-FTIR PM Analysis
| Category | Item | Specifications | Function/Purpose |
|---|---|---|---|
| Sampling Materials | Quartz fiber filters | 47 mm diameter, pre-baked | PM collection for organic analysis |
| Teflon filters | 47 mm diameter | PM collection for ATR-FTIR analysis | |
| Filter cassettes | Size-selective inlets (PM₁₀, PM₂.₅) | Particle size segregation | |
| ATR-FTIR Accessories | ATR crystal | Diamond, Germanium, or ZnSe | Internal Reflection Element (IRE) |
| Pressure tower | FTIR-compatible | Ensures sample contact with crystal | |
| Cleaning solvents | HPLC-grade methanol, acetone | Crystal cleaning between samples | |
| Calibration Standards | Potassium bromide (KBr) | FTIR grade | Background reference |
| Sulfate standards | (NH₄)₂SO₄, analytical grade | Quantification calibration | |
| Nitrate standards | NH₄NO₃, analytical grade | Quantification calibration | |
| Organic standards | n-Alkanes, polyaromatic hydrocarbons | Functional group identification | |
| Data Analysis Tools | Chemometric software | PCA, PLS algorithms | Multivariate spectral analysis |
| Spectral libraries | Custom environmental databases | Compound identification |
The selection of appropriate filter media is particularly critical, as different materials serve complementary purposes. Quartz fiber filters are ideal for subsequent thermal-optical analysis of carbonaceous components, while Teflon filters provide optimal backgrounds for ATR-FTIR analysis despite some interference in the 1270-1050 cm⁻¹ region [38]. The ATR crystal choice (typically diamond for durability or germanium for high refractive index) depends on sample characteristics and required penetration depth [40].
Integration with Broader Research Initiatives
ATR-FTIR analysis of PM contributes significantly to broader environmental research frameworks, including source apportionment studies, health impact assessments, and climate change investigations. The technique has been successfully integrated into established monitoring networks such as the Interagency Monitoring of Protected Visual Environments (IMPROVE), which was created to monitor aerosol composition in Class I areas (National Parks and Wilderness Areas) as mandated by the 1977 Clean Air Act Amendments [6].
The functional group information provided by ATR-FTIR bridges important gaps between conventional measurements. While thermal-optical methods provide total organic carbon (OC) data, and GC-MS offers detailed molecular-level information for specific compounds, ATR-FTIR characterizes the broad spectrum of organic functional groups that comprise the majority of unexplained organic aerosol mass [6] [38]. This comprehensive characterization is particularly valuable for understanding secondary organic aerosol formation through atmospheric processing of precursor gases.
Recent advances have demonstrated the capability of ATR-FTIR to characterize the weathering state of environmental microplastics in freshwater systems, expanding its application beyond traditional air pollution studies [3]. The calculation of specific indexes (Carbonyl Index, Hydroxyl Index, Carbon-Oxygen Index) provides quantitative metrics for tracking polymer degradation under environmental conditions [3]. This capability positions ATR-FTIR as a versatile tool for investigating emerging contaminants in multiple environmental compartments.
ATR-FTIR spectroscopy represents a powerful analytical approach for comprehensive characterization of airborne particulate matter, offering unique capabilities for simultaneous identification of organic functional groups and inorganic ions in environmental samples. The technique's minimal sample preparation requirements, non-destructive nature, and compatibility with complex environmental matrices make it particularly valuable for both research and monitoring applications. While method performance is optimized at higher PM concentrations typically encountered during pollution episodes, ATR-FTIR remains a robust screening tool across concentration ranges.
The integration of ATR-FTIR into broader air quality research frameworks enhances our understanding of aerosol sources, transformation processes, and potential health impacts. As demonstrated in field studies across diverse environments, from Italian freshwater systems to Chinese megacities, this technique provides crucial data linking aerosol composition to emission sources and atmospheric processing. Continued refinement of spectral libraries, calibration approaches, and chemometric data processing will further strengthen the application of ATR-FTIR in addressing complex environmental challenges associated with airborne particulate matter.
Fourier Transform Infrared (FTIR) spectroscopy stands as a cornerstone technique in the identification of organic pollutants, providing molecular fingerprinting capabilities through the excitation of vibrational modes in chemical bonds [8]. However, the analysis of complex environmental mixtures and degradation products presents significant challenges that often surpass the capabilities of standalone FTIR. To address these limitations, advanced hybrid configurations that couple FTIR with complementary analytical techniques have emerged as powerful solutions for comprehensive material characterization and pollutant identification. These integrated systems provide a synergistic analytical approach that delivers more detailed information than any single technique could offer independently.
The evolution of FTIR into hybrid configurations such as TGA-IR and GC-IR represents a significant advancement in analytical capabilities for environmental research and drug development. By combining thermal analysis or chromatographic separation with infrared detection, researchers can overcome the limitation of analyzing multi-component systems where spectral signatures overlap and obscure identification. Within the broader context of FTIR identification of organic pollutants research, these configurations enable not only the detection of pollutants but also the understanding of their thermal behavior, degradation pathways, and composition in complex matrices [41] [8]. This technical guide explores the principles, methodologies, and applications of these advanced configurations, providing researchers with the knowledge to leverage these powerful tools for solving complex analytical challenges.
TGA-IR Configuration: Thermal Degradation Analysis
System Fundamentals and Operating Principles
Thermogravimetric Analysis coupled with Fourier Transform Infrared Spectroscopy (TGA-IR) is an advanced evolved gas analysis technique that integrates the quantitative weight measurement capabilities of TGA with the chemical identification power of FTIR [41]. This configuration provides comprehensive information on a material's thermal stability and decomposition behavior while simultaneously identifying the gases evolved during thermal degradation. The TGA technique quantifies a sample's weight change as a function of temperature or time, subjecting the sample to a precisely controlled temperature program while continuously monitoring its weight [41]. This enables the determination of critical information related to a material's thermal stability and decomposition behavior.
The gases that evolve from the sample through evaporation or thermal decomposition during the TGA measurement are transferred via a heated transfer line through an IR gas cell to be measured with IR spectroscopy [41]. IR spectroscopy uses the interaction between infrared light and the molecular bonds within the evolved gas, which produces a unique spectrum for each type of material. The resulting spectrum reveals valuable details about the functional groups within the evolving gases, aiding in the identification of chemical bonds, molecular structure, and functional groups of the original sample [41]. This combination is particularly valuable for studying the thermal degradation of various polymers, which gives information about their use in various applications, and for identifying components like plasticizers and additives in polymers [41].
Experimental Protocol for TGA-IR Analysis
A standardized experimental protocol for TGA-IR analysis ensures consistent and reliable results:
Sample Preparation: Place 10-20 mg of solid or liquid sample into an open alumina or platinum crucible. For homogeneous powders, ensure representative sampling. For larger particles, gentle grinding may be necessary to improve thermal consistency, though this may not be appropriate for all materials.
Instrument Setup: Configure the TGA with a temperature program appropriate for the material being analyzed. Typical programs include ramped increases of 10-20°C/min from ambient to 800-1000°C under nitrogen or air atmosphere at a flow rate of 50-100 mL/min. Maintain the transfer line temperature at 200-300°C to prevent condensation of evolved gases.
FTIR Configuration: Set the IR spectrometer to collect spectra continuously throughout the experiment with a resolution of 4 cm⁻¹. Collect background spectra before sample heating begins. The IR gas cell should be maintained at an elevated temperature (typically 200-250°C) to prevent analyte condensation.
Data Collection: Initiate the temperature program simultaneously with spectral collection. The TGA records weight loss while FTIR collects spectra of evolved gases at predetermined intervals (typically every 10-20 seconds).
Data Analysis: Correlate specific weight loss events in the TGA curve with the FTIR spectra collected at corresponding times. Use spectral libraries to identify the functional groups and specific compounds associated with each thermal decomposition stage.
Table 1: Key TGA-IR Operational Parameters for Pollutant Analysis
| Parameter | Typical Setting | Notes |
|---|---|---|
| Sample Mass | 10-20 mg | Smaller for materials with high volatile content |
| Temperature Range | 35°C to 900°C | Dependent on material thermal stability |
| Heating Rate | 10-20°C/min | Slower rates improve separation of decomposition events |
| Purge Gas | N₂ or Air | N₂ for pyrolysis, air for oxidative degradation |
| Transfer Line Temperature | 200-300°C | Must be above condensation point of evolved gases |
| IR Spectral Resolution | 4 cm⁻¹ | Balance between sensitivity and scan speed |
| Gas Cell Temperature | 200-250°C | Prevents condensation of analytes |
Application in Organic Pollutant Research
TGA-IR has proven particularly valuable in polyurethane characterization, identifying the building blocks of polyurethanes, which can be valuable for material development and understanding their environmental degradation products [41]. Additionally, it has been applied to ink formulation analysis, determining the composition of unknown cured ink formulations, which may contain hazardous components that leach into the environment [41]. The technique also enables identification of plasticizers and additives in polymers, which is essential for understanding polymer properties and their potential for releasing harmful substances during use or disposal [41].
Diagram 1: TGA-IR System Workflow for evolved gas analysis
GC-IR Configuration: Separation and Identification
System Fundamentals and Operating Principles
Gas Chromatography coupled with Infrared Spectroscopy (GC-IR) combines the superior separation power of GC with the molecular identification capabilities of FTIR. In this configuration, complex mixtures are first separated into individual components by the GC system before being directed to the IR detector for identification. In GC, the gases (which are likely to be mixtures of various components) pass through a separation column, separating the individual components based on their volatility and column affinity [41]. This separation is crucial for analyzing complex mixtures where component spectra would otherwise overlap and obscure identification.
After chromatographic separation, the eluting components are transferred to the IR spectrometer for analysis. The interface between the GC and FTIR typically consists of a heated light pipe or specialized flow cell maintained at elevated temperatures to prevent analyte condensation. As the separated components pass through the IR beam, they produce characteristic absorption spectra that serve as molecular fingerprints. The resulting infrared spectrum provides a detailed chemical fingerprint, enabling comprehensive compositional analysis [8]. This configuration is particularly powerful for identifying volatile and semi-volatile organic compounds in complex matrices, making it invaluable for environmental pollutant analysis and pharmaceutical development.
Experimental Protocol for GC-IR Analysis
A standardized protocol for GC-IR analysis of organic pollutants includes:
Sample Preparation: Prepare liquid samples or extracts in appropriate volatile solvents (dichloromethane, hexane) compatible with GC analysis. Filter or concentrate samples as needed to achieve appropriate detection limits. For solid samples, employ appropriate extraction techniques prior to analysis.
GC Configuration: Install a capillary column appropriate for the target analytes (typically 30m × 0.25mm × 0.25μm). Set the injector temperature 10-20°C above the highest boiling point component. Program the oven temperature to ramp from 40°C (hold 2 min) to 300°C at 10-15°C/min. Use helium as carrier gas at 1.0 mL/min constant flow.
FTIR Interface Setup: Maintain the light pipe or flow cell at 250-300°C. Set the IR spectrometer to collect spectra at 4-8 cm⁻¹ resolution with rapid scan capability (typically 1-2 spectra per second). Align the optical system for maximum throughput.
Data Collection: Inject 1-2 μL of sample in split or splitless mode depending on concentration. Simultaneously trigger GC data collection and IR spectral acquisition. Monitor the total ion chromatogram while continuously collecting IR spectra.
Data Analysis: Use the Gram-Schmidt reconstruction to detect eluting components. Interrogate individual component spectra against commercial libraries. For unknown compounds, analyze functional groups based on characteristic absorption frequencies.
Table 2: Key GC-IR Operational Parameters for VOC Analysis
| Parameter | Typical Setting | Notes |
|---|---|---|
| Injection Volume | 1-2 μL | Split/splitless based on concentration |
| Column Type | 30m × 0.25mm × 0.25μm | Mid-polarity stationary phase preferred |
| Oven Program | 40°C to 300°C at 10-15°C/min | Optimized for separation efficiency |
| Carrier Gas | Helium at 1.0 mL/min | Constant flow mode |
| Light Pipe Temperature | 250-300°C | Prevents analyte condensation |
| IR Scan Rate | 1-2 spectra/second | Balances temporal and spectral resolution |
| Spectral Resolution | 4-8 cm⁻¹ | Optimized for vapor phase spectra |
Application in Organic Pollutant Research
GC-IR has been successfully applied to monitoring Volatile Organic Compounds (VOCs) in air, with studies demonstrating detection capabilities down to part per million (ppm) levels for compounds like styrene, acetone, ethanol, and isopropanol [10]. The technique enables both the detection of VOCs at very low concentrations and their reliable discrimination, which is crucial for assessing environmental and health risks [10]. GC-IR also plays a vital role in assessing organic pollutants desorbed from plastic litter in marine environments, where it helps identify compounds such as fragrances, insect repellents, and UV filters that accumulate on plastic debris [28]. When combined with mass spectrometry in TGA-IR-GC-MS systems, the technique provides even greater analytical power for identifying components released during thermal degradation of materials, providing further insight into the original composition of the material [41].
Integrated TGA-IR-GC-MS: A Comprehensive Solution
System Architecture and Workflow
The most advanced configuration in evolved gas analysis integrates TGA, IR, and GC-MS into a single comprehensive analytical system. This powerful synergy provides unprecedented capability for characterizing complex materials and identifying organic pollutants. In this setup, the TGA instrument quantifies weight changes during controlled heating, with the evolved gases first directed through an FTIR spectrometer for initial functional group identification, and then to a GC-MS system for precise separation and definitive identification of individual components [41]. The TGA-IR-GC-MS analysis is an analytical method that integrates three powerful techniques that together form an analytical synergy providing comprehensive information on a material's chemical properties [41].
The operational workflow begins with the TGA module, where the sample undergoes precisely controlled thermal programming while weight changes are continuously monitored. The gases evolving from the sample at specific temperatures during the TGA measurement can be collected to inject into the GC-MS [41]. In the GC phase, the gaseous mixtures pass through a separation column, separating the individual components based on their volatility and column affinity [41]. Finally, MS analyzes these separated components, providing information regarding their molecular mass and fragmentation patterns [41]. This multi-technique approach helps identify the components released during thermal degradation of the material, providing further insight into the original composition of the material [41].
Experimental Protocol for TGA-IR-GC-MS Analysis
The integrated nature of TGA-IR-GC-MS requires careful method development:
System Configuration: Connect the TGA, FTIR, and GC-MS using heated transfer lines maintained at temperatures appropriate for the target analytes (typically 250-300°C). Place cryogenic traps or adsorption tubes between the TGA and GC-MS to focus specific evolution periods if needed.
TGA Method Development: Establish temperature programs based on preliminary TGA data. Identify key weight loss regions for detailed analysis. Program multiple heating rates or isothermal holds to separate overlapping decomposition events.
FTIR Method Setup: Configure the FTIR to collect spectra continuously throughout the experiment. Set appropriate spectral range and resolution (typically 4000-600 cm⁻¹ at 4 cm⁻¹ resolution). Establish a library of reference spectra for expected decomposition products.
GC-MS Method Development: Select appropriate GC column and temperature program based on the volatile compounds identified in preliminary FTIR analysis. Optimize MS parameters including ionization energy, mass range, and scan rate.
Data Integration and Analysis: Correlate data from all three techniques by aligning temporal events. Use the FTIR data for initial functional group identification and the GC-MS data for definitive compound identification and quantification.
Diagram 2: Integrated TGA-IR-GC-MS analysis pathways
Advanced Data Analysis Techniques
Spectral Interpretation and Functional Group Identification
Effective interpretation of FTIR spectra from TGA-IR and GC-IR systems requires a systematic approach to identifying characteristic functional group absorptions. The FTIR spectrum provides a detailed chemical fingerprint, enabling comprehensive compositional analysis through careful analysis of the infrared absorption spectra from a sample, which discerns molecular vibrations that demonstrate the presence of designated functional groups [8]. The resulting spectrum is represented as a two-dimensional graph, in which the axes represent the intensity and frequency of sample absorption [8].
For environmental pollutant analysis, certain spectral regions are particularly informative. The region between 4000-1300 cm⁻¹ is known as the functional group region, where characteristic stretches occur that can identify major chemical classes. The region from 1300-400 cm⁻¹ constitutes the fingerprint region, where complex vibration patterns provide unique identifiers for specific compounds. When analyzing mixtures, advanced approaches such as multicomponent searching are essential, as traditional subtraction methods can lead to erroneous results, especially with totally absorbing peaks or when environmental changes alter peaks slightly [23].
Table 3: Key FTIR Absorption Frequencies for Organic Pollutant Identification
| Peak Position (cm⁻¹) | Functional Group | Compound Class | Pollutant Examples |
|---|---|---|---|
| 3300-3400 | N-H stretching | Primary amine | Aromatic amines |
| 2500-3300 | O-H stretching | Carboxylic acid | Organic acids |
| 2222-2260 | C≡N stretching | Nitrile | Industrial chemicals |
| 1690-1740 | C=O stretching | Aldehydes, Ketones, Esters | Carbonyl-containing VOCs |
| 1660-1680 | C=O stretching | Secondary amide | Pharmaceutical residues |
| 1600-1620 | C=C stretching | Aromatic compounds | PAHs |
| 1500-1550 | N-O stretching | Nitro compounds | Explosives residues |
| 1000-1400 | C-F stretching | Fluoro compound | Fluorinated pollutants |
Multivariate Analysis and Machine Learning Approaches
The complex spectral data generated by TGA-IR and GC-IR systems often requires advanced computational approaches for meaningful interpretation. Random forests, a machine learning technique for classification and regression, has proven particularly useful for interrogating high dimensional datasets like FTIR spectral data [42]. Random forests can be used on large datasets because random sub-sampling allows trees to grow without suffering from overfitting [42]. This capability is especially valuable when dealing with complex environmental samples where multiple pollution sources contribute to the overall composition.
Other powerful approaches include the use of spectral derivatives and smoothing algorithms to enhance classification accuracy. Studies have demonstrated that processing FTIR spectra using Savitzky-Golay smoothing on data derivatives can enhance classification accuracy when using k-nearest neighbour classification [42]. For homogeneous mixtures, cumulative searching algorithms that don't rely on spectral subtraction have shown superior performance compared to traditional approaches, as they avoid problems with derivative-shaped bands or totally absorbing peaks that plague subtraction methods [23]. These advanced data processing techniques are essential for unlocking the full potential of TGA-IR and GC-IR configurations for complex mixture analysis.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Essential Materials and Reagents for TGA-IR and GC-IR Analysis
| Item | Function | Application Notes |
|---|---|---|
| High-Purity Inert Gases (N₂, He) | TGA purge gas, GC carrier gas | Essential for preventing oxidation in TGA; He preferred for GC-MS |
| Certified VOC Standard Mixtures | System calibration and quantification | Required for method validation and quantitative analysis |
| FTIR Calibration Standards | Wavelength and intensity verification | Polystyrene films for routine verification |
| Sorbent Tubes (Tenax, Charcoal) | Pre-concentration of volatile analytes | Extends detection limits for trace-level pollutants |
| Deuterated Internal Standards | Quantification in GC-MS | Corrects for matrix effects and preparation losses |
| Spectral Library Databases | Compound identification | Commercial and custom libraries for specific applications |
| Heated Transfer Lines | Maintain gas phase analytes | Prevent condensation between system components |
| High-Temperature GC Columns | Separation of complex mixtures | Mid-polarity stationary phases for broad applicability |
TGA-IR and GC-IR configurations represent significant advancements in the analytical capability to identify and characterize organic pollutants in complex mixtures. By combining the separation and thermal analysis capabilities of GC and TGA with the molecular specificity of FTIR, these hybrid techniques provide researchers with powerful tools for understanding pollutant composition, behavior, and transformation in environmental systems. The flexibility of the whole evolved gas system offers several combinations of techniques including TGA-IR, TGA-MS, TGA-GC-MS and TGA-IR-GC-MS analyses, making it possible to select the best combination of techniques to answer the material questions [41].
As environmental pollution becomes increasingly complex, these advanced configurations will play an ever more critical role in monitoring, understanding, and mitigating the impact of organic pollutants on ecosystems and human health. The continued development of these techniques, coupled with advanced data analysis approaches including machine learning, promises to further enhance our ability to unravel the complexity of environmental contamination and develop effective remediation strategies. For researchers focused on FTIR identification of organic pollutants, mastery of these advanced configurations provides a significant competitive advantage in addressing the analytical challenges of modern environmental science.
Overcoming Analytical Challenges: A Guide to Optimizing FTIR Results
Fourier-Transform Infrared (FTIR) spectroscopy stands as a powerful tool for identifying and quantifying organic pollutants in environmental research, enabling real-time monitoring of hazardous air pollutants as mandated by clean air regulations [43] [44]. However, the analytical pathway from sample collection to definitive identification is fraught with technical challenges that can compromise data integrity. Two of the most pervasive obstacles are spectral interference from atmospheric water vapor and the physical limitations imposed by sample opacity. Water vapor produces strong, broad absorption bands that can obscure the characteristic spectral fingerprints of target analytes [45]. Simultaneously, improper handling of opaque or highly absorbing samples can lead to phenomena like total absorbance, yielding non-linear data and poor spectral quality [46]. This guide details advanced methodologies and practical protocols to navigate these pitfalls, ensuring the reliability of FTIR data in critical drug development and environmental research.
Core Challenge 1: Water Vapor Interference
Water vapor interference arises because the vibrational frequencies of water molecules—specifically OH stretching (2700–3700 cm⁻¹) and bending (around 1650 cm⁻¹)—fall squarely within the mid-IR region used for analysis [46]. These bands are broad and intense, often overlapping with and obscuring the signals of organic functional groups. In quantitative work, this interference introduces significant uncertainty, particularly in the regions of 1325–1900 cm⁻¹ and 3550–3950 cm⁻¹, which the National Institute of Standards and Technology (NIST) explicitly flags as compromised for analysis due to water absorption [43].
Experimental Protocol for Mitigating Water Interference
A robust, multi-layered approach is required to mitigate water interference effectively. The following protocol is designed for extractive FTIR analysis of gas-phase pollutants.
Step 1: Instrument Purge and Atmosphere Control
- Employ a purge system with dry, inert gas such as nitrogen or zero air to displace ambient moisture from the optical path of the FTIR instrument [45].
- Continuously purge the spectrometer and the external sampling cell during both background and sample measurements.
- Maintain a stable temperature in the laboratory and for the sampling cell to prevent condensation [45].
Step 2: Acquisition of Atmospheric Background Spectra
- Collect multiple background spectra using an empty, purged gas cell, or by sampling purified nitrogen or dry, pollutant-free air from the measurement location.
- For dynamic field measurements, record background spectra frequently throughout the experiment to account for temporal changes in atmospheric humidity [47].
Step 3: Data Acquisition and Advanced Spectral Processing
- Collect the sample spectrum. For quantitative analysis based on databases like the NIST Quantitative Infrared Database, ensure your instrumental resolution and apodization functions match the reference data as closely as possible [43].
- Utilize advanced software tools like VaporFit, an open-source application that employs a multispectral least-squares approach. Instead of relying on a single background subtraction, VaporFit automatically optimizes subtraction coefficients based on multiple atmospheric measurements, providing superior correction for variable water vapor and CO₂ [47].
Step 4: Validation of Correction
- Visually inspect the corrected spectrum for the characteristic residual derivative-shaped features that indicate improper subtraction.
- Use built-in software metrics, such as objective smoothness measures or principal component analysis (PCA) modules, to objectively evaluate the quality of the atmospheric correction [47].
Quantitative Impact of Water Vapor
The following table summarizes the key spectral regions affected by common atmospheric interferents, including water vapor, which must be recognized and corrected for accurate analysis.
Table 1: Primary Atmospheric Interferents in FTIR Spectroscopy
| Interferent | Spectral Regions (cm⁻¹) | Impact on Pollutant Analysis |
|---|---|---|
| Water Vapor (H₂O) | 1325 – 1900, 3550 – 3950 [43] | Obscures a wide range of C=O, C=C, C-H, and O-H stretches in organic pollutants. |
| Carbon Dioxide (CO₂) | 2295 – 2385 [43] | Can interfere with nitrile (C≡N) and certain triple-bond absorptions. |
| Carbon Monoxide (CO) | 2050 – 2225 [43] | May overlap with acetylene and other less common triple-bonded pollutants. |
Water Vapor Mitigation Workflow
Core Challenge 2: Sample Opacity and Total Absorbance
Sample opacity leads to the problem of total absorbance, where a sample is too thick, too concentrated, or too highly scattering for the IR beam to penetrate effectively. This results in saturated, non-linear peaks that are useless for quantification and can mask weaker absorption features from trace pollutants [46]. The choice of sampling technique is critical to overcoming this physical limitation.
Experimental Protocol for Opaque and Scattering Samples
The appropriate sampling technique depends on the physical state of the pollutant and the analytical question.
Protocol A: Attenuated Total Reflectance (ATR) for Solids and Liquids
- Principle: The IR beam travels through a high-refractive-index crystal, generating an evanescent wave that penetrates 0.8–2.0 µm into the sample in contact with the crystal. This minimal penetration avoids total absorbance [48] [46].
- Methodology: Place the solid or liquid sample directly onto the ATR crystal (e.g., diamond, ZnSe). Apply firm, consistent pressure to ensure good contact. Acquire the spectrum with minimal preparation. ATR is ideal for analyzing filtered particulate matter, dried sludge, or concentrated liquid wastes [48].
- Consideration: ATR spectra show intensity differences compared to transmission spectra but are easily corrected by software [46].
Protocol B: Transmission with Gas Cells for Gaseous Pollutants
- Principle: The IR beam passes through a sealed cell containing the gas sample. The pathlength is critical; too long a path with a high-concentration gas will lead to total absorbance [48].
- Methodology: For low-concentration pollutants (e.g., ambient air monitoring), use a long-path gas cell (e.g., 20 m). For high-concentration stack emissions, a short-path cell is appropriate. The cell should be sealed with IR-transparent windows (e.g., BaF₂, ZnSe) and can be heated to prevent condensation [48].
Protocol C: Diffuse Reflectance (DRIFTS) for Powders and Soils
- Principle: Incident IR light is scattered by powdered samples, and the diffusely reflected light is collected. This is ideal for highly scattering solid samples like soils, catalysts, or powdered sediments [48].
- Methodology: Finely grind the sample and mix it with a non-absorbing powder like KBr to reduce absorption and minimize specular reflection. Loosely pack the mixture into a sample cup. A Kubelka-Munk correction applied to the collected spectrum allows for quantitative comparison with transmission libraries [48].
The Researcher's Toolkit for Sampling
Selecting the right accessory is paramount for managing sample opacity. The table below catalogs essential solutions for various sample types.
Table 2: Essential FTIR Sampling Techniques & Reagents for Pollutant Analysis
| Technique | Best For Pollutant Type | Key Research Reagents & Materials | Function & Rationale |
|---|---|---|---|
| ATR | Solid residues, viscous liquids, microplastics | Diamond, ZnSe, or Germanium crystal | Rugged, chemically inert surface for minimal sample preparation. |
| Transmission (Gas Cell) | Volatile Organic Compounds (VOCs), stack gases | Sealed gas cell with BaF₂ or ZnSe windows; long-path (≥10m) for low conc. | Provides controlled pathlength to avoid signal saturation for gases [48]. |
| KBr Pellets | Powdered solids, soils | Potassium Bromide (KBr) | IR-transparent matrix to dilute strongly absorbing samples [48]. |
| DRIFTS | Highly scattering powders, soils, catalysts | KBr or KCl powder | Diluent that reduces absorption and shifts analysis to diffuse reflectance [48]. |
Sampling Technique Selection
Advanced Data Analysis for Complex Mixtures
Environmental samples are rarely pure; they are complex mixtures where spectral peaks from multiple components overlap. Traditional analysis by sequential library search and subtraction is prone to error, especially with poor water correction or totally absorbing peaks [23].
Protocol for Multicomponent Spectral Searching
- Methodology: Use software capable of true multicomponent searching (e.g., OMNIC Specta, ACD/Labs Spectrus). Instead of subtracting one component at a time, the algorithm tests linear combinations of library spectra to find the set that best matches the entire unknown spectrum simultaneously [23].
- Interpretation: The result is a composite spectrum and a list of candidate constituents. This method is highly effective for gas-phase mixtures, which behave nearly ideally [23]. Critical Note: The composite percentages from normalized libraries do not directly reflect true concentration; they are a qualitative guide. For quantification, use unnormalized reference spectra with known absorptivities [23].
- Assessing Match Quality: Rely not only on the Hit Quality Index (HQI) of the top match but also on the "gap" or difference in HQI between successive hits. A significant gap between the first and second hit suggests a unique match, while a small gap indicates a cluster of similar compounds, signaling the need for careful review of the top candidates [49].
Successfully leveraging FTIR spectroscopy for organic pollutant identification demands a rigorous, multi-faceted approach that confronts the technique's inherent vulnerabilities head-on. By implementing controlled purge systems and advanced computational corrections like VaporFit, researchers can effectively neutralize the confounding effects of atmospheric water vapor. Simultaneously, a strategic understanding of sampling techniques—from the simplicity of ATR to the specialized application of DRIFTS and gas cells—empowers analysts to transform opaque, intractable samples into high-quality spectral data. When combined with robust multicomponent analysis that moves beyond simplistic library searches, these protocols form a comprehensive defense against common pitfalls. This rigorous framework ensures that FTIR data remains a trustworthy pillar in environmental monitoring, regulatory compliance, and public health protection.
Fourier Transform Infrared (FTIR) spectroscopy has become an indispensable technique for the identification and analysis of organic pollutants, providing molecular fingerprinting capabilities essential for environmental research. The technique's effectiveness hinges on the precise optimization of instrumental parameters and the strategic selection of sampling accessories, which together determine the sensitivity and resolution of the acquired spectra. In the context of identifying organic pollutants—a critical application for safeguarding public and environmental health—proper configuration ensures reliable detection of trace-level contaminants and accurate differentiation between chemically similar compounds [8]. This technical guide provides a comprehensive framework for method development, focusing on the interplay between parameter tuning and accessory selection to maximize analytical performance in organic pollutant research.
Core Principles of FTIR Sensitivity and Resolution
Fundamental Definitions and Relationships
In FTIR spectroscopy, sensitivity refers to the ability to detect weak absorption bands, which is crucial for identifying low-concentration organic pollutants. It is directly influenced by the signal-to-noise ratio (SNR), which can be improved by increasing the number of scans, optimizing optical throughput, and selecting appropriate detectors [50]. Resolution, defined as the smallest spectral separation between two distinguishable bands, determines the technique's ability to reveal fine structural details and separate overlapping peaks from complex mixtures [51]. For organic pollutant analysis, high resolution is particularly valuable for distinguishing between homologous compounds or isomers with subtle spectral differences.
The relationship between sensitivity and resolution involves inherent trade-offs. Higher spectral resolution (achieved by increasing the optical path difference) typically reduces energy throughput to the detector, potentially degrading SNR unless compensated by longer acquisition times [50]. Understanding this balance is essential for optimizing FTIR methods for specific analytical challenges in environmental monitoring.
FTIR in Organic Pollutant Identification
FTIR identifies organic pollutants through their characteristic vibrational absorption patterns, creating unique molecular fingerprints across the mid-infrared region (4000-400 cm⁻¹) [8]. The technique can probe various sample matrices, including water, soils, and biological tissues, making it versatile for environmental analysis. Functional groups common in organic pollutants—such as carbonyls in pesticides, aromatic rings in polycyclic aromatic hydrocarbons, and C-Cl bonds in chlorinated solvents—produce distinctive absorption bands that enable both identification and, with proper calibration, quantification [8] [52].
Parameter Tuning for Enhanced Performance
Resolution Optimization
Spectral resolution is primarily controlled by the maximum optical path difference (OPD) in the interferometer. Higher resolutions require longer OPDs, which improve the ability to distinguish closely spaced spectral features but demand greater mechanical stability and longer acquisition times [50].
Practical Resolution Guidelines:
- 4 cm⁻¹: Suitable for routine analysis of organic pollutants with well-separated absorption bands.
- 2-1 cm⁻¹: Recommended for complex mixtures or when identifying pollutants with similar chemical structures.
- <1 cm⁻¹: Typically reserved for gas-phase analysis or research requiring the highest level of spectral detail [50].
Instrument-to-instrument variation in peak position accuracy is typically within 1.1 cm⁻¹ for resolutions of 4 cm⁻¹ or higher, increasing to 4.7 cm⁻¹ at 16 cm⁻¹ resolution [51]. This positional accuracy is critical for reliable library matching and identification of unknown organic pollutants.
Table 1: Effect of Spectral Resolution on Wavenumber Accuracy
| Spectral Resolution (cm⁻¹) | Typical Wavenumber Accuracy (cm⁻¹) | Recommended Applications for Organic Pollutant Analysis |
|---|---|---|
| 32 | 10.4 | Preliminary screening of strong, isolated bands |
| 16 | 4.7 | Qualitative analysis of simple mixtures |
| 8 | 2.2 | General purpose environmental analysis |
| 4 | 1.1 | Detailed analysis of complex environmental samples |
| 2 | <1.0 | High-resolution studies of gaseous pollutants |
| 1 | <0.5 | Research-grade analysis requiring finest spectral details |
Scan Number and Signal Averaging
The number of scans averaged directly impacts the signal-to-noise ratio through the relationship SNR ∝ √N, where N is the number of scans. This fundamental principle makes scan number optimization essential for detecting trace-level organic pollutants [53].
Experimental Protocol for Scan Number Optimization:
- Preliminary Acquisition: Collect successive spectra at varying scan numbers (e.g., 10, 20, 30, 40, 50, 60, 70, 80, 90, 100) from a representative sample [53].
- Similarity Assessment: Calculate the Standardized Moment Distance Index (SMDI) between replicate measurements to quantify spectral stability [53].
- Threshold Determination: Identify the point where additional scans no longer significantly improve spectral similarity (stability plateau).
- Validation: Verify that the selected scan number provides sufficient SNR for detecting target pollutant bands without excessively prolonging analysis time.
Research indicates that for soil samples (a common matrix for organic pollutant analysis), spectral stability improves markedly up to approximately 50 scans, with diminishing returns beyond this point [53]. The optimal value depends on the specific matrix and analyte concentration, necessitating empirical determination for each application.
Apodization and Phase Correction
Apodization functions reduce spectral artifacts (sidelobes) that arise from the finite nature of the interferogram but inevitably broaden spectral features. The choice of apodization function represents a compromise between resolution and SNR [50]. Common functions include:
- Happ-Genzel: General-purpose choice offering good balance
- Norton-Beer Medium: Provides enhanced resolution for closely spaced bands
- Boxcar: Maximizes resolution but increases sidelobe artifacts
Phase correction algorithms (Mertz or Forman) ensure the interferogram is correctly transformed into a single-beam spectrum, critical for quantitative accuracy when analyzing pollutant concentrations [50].
Accessory Selection for Organic Pollutant Analysis
Guide to Accessory Selection by Sample Type
The sampling accessory must be matched to the physical state and properties of the environmental sample containing organic pollutants. Different accessories enable analysis of various sample types while maximizing sensitivity and spectral quality [54].
Table 2: FTIR Accessory Selection Guide for Environmental Samples
| Sample Type | Recommended Accessory | Key Considerations for Organic Pollutant Analysis |
|---|---|---|
| Liquid Pollutants | Horizontal ATR with diamond crystal | Minimal preparation; ideal for aqueous samples |
| (Volatile organic solvents) | Sealed liquid cell with KBr or CaF₂ windows | Prevents evaporation; suitable for quantitative analysis |
| (Oil and hydrocarbon spills) | Demountable liquid cell or single-bounce ATR | Handles viscous samples without dilution |
| Solid Pollutants | Single-bounce ATR (diamond, ZnSe) | Minimal preparation; good for powders, soils, and sediments |
| (Powders, soils, sediments) | Diffuse Reflectance (DRS-8000A) mixed with KBr powder | Enhanced sensitivity for diluted samples |
| (Plastics, polymers) | Multi-bounce ATR or transmission with film holder | Adapts to sample thickness and transparency |
| Gaseous Pollutants | Long-path gas cell (≥10 m optical path) | Low detection limits (0.1-1 ppm) for air quality monitoring |
Attenuated Total Reflectance (ATR) Techniques
ATR has become the predominant sampling technique for organic pollutant analysis due to its minimal sample preparation requirements and compatibility with a wide range of sample types. The technique relies on total internal reflection, generating an evanescent wave that penetrates 0.5-5 µm into the sample, depending on the crystal material, wavelength, and angle of incidence [54].
Crystal Material Selection:
- Diamond: Chemically inert, durable, suitable for hard particles and aggressive chemicals; penetration depth ~2 µm at 1000 cm⁻¹
- Zinc Selenide (ZnSe): Wider spectral range than diamond; not suitable for acidic samples or low pH aqueous solutions
- Germanium (Ge): Shallow penetration depth (~0.5-0.8 µm); ideal for strongly absorbing samples or surface analysis
For most environmental applications involving organic pollutants, diamond ATR provides the best combination of durability, spectral range, and sensitivity.
Integrated Method Development and Workflow
Experimental Design for Organic Pollutant Analysis
A systematic approach to method development ensures optimal sensitivity and resolution for specific analytical challenges in organic pollutant identification.
Diagram 1: FTIR Method Development Workflow
Advanced Resolution Enhancement Techniques
When hardware limitations restrict achievable resolution, computational methods can effectively enhance spectral detail:
Fourier Self-Deconvolution
- Algorithmically narrows bandwidth by deconvoluting the instrument line shape function
- Reveals underlying components in overlapping bands
- Requires careful parameter selection to avoid artifact generation
Derivative Spectroscopy
- Second-derivative processing emphasizes sharp peaks and suppresses broad features
- Enhances resolution of overlapping bands
- Amplifies noise, requiring prior smoothing
Multivariate Curve Resolution
- Chemometric approach for extracting pure component spectra from complex mixtures
- Particularly valuable for unresolved pollutant mixtures in environmental samples
Research Reagent Solutions for Method Validation
Table 3: Essential Materials and Reagents for FTIR Analysis of Organic Pollutants
| Reagent/Material | Specification | Application in Organic Pollutant Analysis |
|---|---|---|
| ATR Crystals | Diamond, ZnSe, or Ge crystals | Sample presentation with minimal preparation |
| KBr Powder | FTIR-grade, purity >99% | Preparation of pellets for transmission measurements |
| Solvent Standards | HPLC-grade solvents (acetonitrile, hexane) | Extract organic pollutants from environmental matrices |
| Reference Materials | NIST-traceable polystyrene film | Instrument performance validation and wavelength calibration |
| Sealed Liquid Cells | Fixed pathlength (0.1-1.0 mm) with KBr or CaF₂ windows | Quantitative analysis of volatile organic compounds |
| Gas Calibration Standards | Certified ppm-level organic vapors in inert gas | Method validation for gaseous pollutant analysis |
Troubleshooting Common Performance Issues
Addressing Sensitivity Limitations
- Poor Signal-to-Noise Ratio: Increase number of scans; verify detector performance; check mirror alignment; ensure sample is properly positioned in the beam [50] [53].
- Weak Absorption Bands: Concentrate sample if possible; use an accessory with longer effective pathlength (e.g., multiple bounce ATR or transmission cell); confirm beam splitter and detector are appropriate for spectral range of interest.
- Baseline Instability: Allow sufficient instrument warm-up time; control environmental humidity and temperature; purge instrument with dry air to reduce water vapor contributions [55].
Correcting Resolution Problems
- Poorly Resolved Peaks: Verify resolution setting matches analytical requirements; check mirror movement for mechanical issues; ensure apodization function is appropriate for the application [51].
- Wavenumber Accuracy Issues: Regularly calibrate with polystyrene reference standard; monitor for thermal stability problems; check laser frequency calibration [51].
- Spectral Artifacts: Identify and eliminate sources of external vibration; check for interferogram sampling errors; verify phase correction parameters [55].
Optimizing FTIR spectroscopy for organic pollutant identification requires systematic attention to both instrumental parameters and accessory selection. Resolution settings between 1-4 cm⁻¹ typically provide the best balance between spectral detail and practical acquisition times, while scan numbers of 32-64 often yield sufficient signal-to-noise for trace detection without excessive measurement duration. The widespread adoption of ATR accessories has simplified sample analysis across various environmental matrices, though transmission and specialized gas cells remain valuable for specific applications. By implementing the protocols and guidelines presented in this technical guide, researchers can significantly enhance the sensitivity and resolution of their FTIR methods, leading to more reliable identification and characterization of organic pollutants in environmental research.
Fourier Transform Infrared (FTIR) Spectroscopy stands as a cornerstone analytical technique for the identification and characterization of organic pollutants across diverse environmental and research matrices. Its effectiveness stems from the ability to generate unique "fingerprint" infrared spectra for most organic and polymeric compounds, enabling rapid identification of chemical families and specific contaminants [56] [39]. However, a comprehensive understanding of its operational limits—specifically its detection thresholds and inherent surface sensitivity—is paramount for researchers and drug development professionals aiming to generate reliable, interpretable data. This guide delves into the core technical specifications of FTIR, providing a detailed examination of these limitations within the context of organic pollutant research, supported by experimental protocols and quantitative data visualization.
The utility of FTIR in detecting organic pollutants is bounded by two primary categories of limitations: its detection thresholds, which define the minimum amount of material that can be reliably observed, and its surface sensitivity, which determines the depth of the sample that contributes to the measured signal.
Detection Thresholds
FTIR does not detect elements but rather molecular functional groups, and its detection limits are typically expressed in terms of concentration or film thickness [56] [39]. The following table summarizes the key detection parameters:
Table 1: FTIR Detection Thresholds for Analytical Applications
| Parameter | Typical Value | Context and Implications |
|---|---|---|
| Quantitative Detection Limit | 1 - 10 wt% | For quantification of known components in a mixture [56] [39]. |
| Identification Limit | 5 - 20 wt% | For identifying unknown components within a material [56] [39]. |
| Minimum Analysis Area | ~15 - 50 µm | The smallest spot size that can be analyzed; a visible sample is typically analyzable [56] [39]. |
| Lateral Resolution/Probe Size | > 15 - 50 µm | Governs the ability to analyze small, discrete particles or contaminants [56] [39]. |
The Limit of Detection (LOD) can be systematically analyzed and improved. From a statistical perspective, the LOD can be framed as a binary classification problem, defined as the smallest concentration of an analyte that can be distinguished from a blank sample with a specified significance level [57]. This is formalized as:
[ yD = \bar{y}B + k \cdot \sigma_B ]
Where ( yD ) is the smallest detectable signal, ( \bar{y}B ) is the mean blank signal, ( \sigma_B ) is the standard deviation of the blank, and ( k ) is a numerical factor (often 2 or 3) related to confidence levels [57]. For IR imaging, sophisticated spectral analysis approaches—such as using a single wavenumber, averaging over discrete wavenumbers, or calculating the total spectral distance—can further optimize the LOD [57].
Surface Sensitivity and Depth Resolution
FTIR is inherently a surface-sensitive technique, which is a critical consideration when analyzing thin film pollutants or coatings. The depth from which the IR signal is collected varies significantly with the sampling mode.
Table 2: Surface Sensitivity and Sampling Depth in FTIR
| Sampling Mode | Typical Depth Resolution / Information Depth | Technical Basis and Applications |
|---|---|---|
| Attenuated Total Reflectance (ATR) | ~0.1 - 1.0 µm [56] [39] | Depth depends on the wavelength of IR light, the refractive index of the ATR crystal (e.g., Germanium, Diamond), and the sample. Provides excellent surface sensitivity for analyzing thin films and surface contaminants. |
| Transmission FTIR | The entire sample thickness | Infrared light passes through the entire sample, which can be advantageous for bulk analysis but may lead to signal saturation for thick samples [56]. |
The extreme surface sensitivity of specific ATR accessories can be harnessed effectively. For instance, using a Germanium ATR crystal (refractive index of 4.0) to analyze a glass surface (refractive index ~1.5) can yield an information depth of only about 0.5 µm at a wavenumber of 1100 cm⁻¹ [58]. This high surface sensitivity has proven crucial in conservation science for detecting thin gel layers on historic glass that were undetectable by other methods like cross-sectioning and SEM [58].
Experimental Protocols for Pushing the Limits
Protocol: Determining the Limit of Detection for a Protein Contaminant
This protocol, adapted from a rigorous methodological study, outlines how to determine the LOD for a protein like Bovine Serum Albumin (BSA) using FTIR imaging [57].
- Fabricate a Microarray: Create a protein microarray with uniform spots (~2.8 nL volume) covering a wide concentration range (e.g., 0.05 to 10 mg/mL). This provides a standardized sample set.
- Acquire Spectral Data: Scan the microarray spots and blank substrate areas using standard FT-IR imaging parameters (e.g., using a widefield or line scanning system).
- Spectral Analysis and Binary Classification: Frame the LOD as a binary hypothesis test between the sample and blank.
- Calculate the mean absorbance signal (( \bar{x}D )) for each protein concentration and the mean blank signal (( \bar{x}B )).
- Calculate the separation parameter ( k = (\bar{x}D - \bar{x}B) / \sigma ), where ( \sigma ) is the standard deviation of the measurements.
- Employ different spectral analysis methods to optimize ( k ):
- Single Wavenumber Absorbance (SWA): Use the frequency where analyte absorption is highest.
- Discrete Wavenumber Average (DWA): Average the signal over a selected set of frequencies to improve the signal-to-noise ratio.
- Spectral Distance Analysis (SDA): Use the full recorded spectrum to calculate the spectral distance between sample and blank, which can incorporate noise correlation.
- Post-processing: Apply techniques like Minimum Noise Fraction (MNF) transformation to denoise the data, which can lower the practical LOD.
- Establish LOD: The LOD is the lowest concentration where the signal can be reliably distinguished from the blank. Using this methodology, LODs for BSA of approximately 0.12 - 0.16 mg/mL have been achieved under typical imaging conditions, which can be improved to ~0.075 mg/mL with post-processing [57].
Protocol: Assessing Surface Degradation using High-Refractive-Index ATR
This protocol details the use of Germanium ATR to probe surface degradation on glass, a method applicable to studying thin organic films or corrosion layers on surfaces [58].
- Sample Preparation: Obtain clean, flat samples. For complex objects like historic daguerreotype cover glasses, analyze the surface directly without modification.
- Instrument Setup: Configure an FTIR microscope equipped with a Germanium ATR crystal. Germanium provides a very shallow information depth due to its high refractive index.
- Spectral Collection: Acquire spectra from multiple points on the sample surface. For degradation studies, compare spectra from apparently degraded and pristine areas.
- Data Interpretation: Analyze the resulting spectra for changes in functional groups. In the case of glass degradation, the "splitting factor" – the difference in maxima wavenumbers between the Si-O-Si (~1100 cm⁻¹) and Si-O-Na/K (~970 cm⁻¹) bands – can be used to quantify the degree of surface deterioration [58].
The workflow for this surface-sensitive analysis is outlined below:
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table catalogues key materials and accessories critical for experiments designed to address the limits of FTIR spectroscopy.
Table 3: Essential Research Reagents and Materials for Advanced FTIR Analysis
| Item | Function and Application |
|---|---|
| Germanium ATR Crystal | Provides extreme surface sensitivity (~0.5 µm depth) for analyzing ultra-thin films, surface gels, and degradation layers [58]. |
| Diamond ATR Crystal | A robust crystal for general-purpose ATR analysis of a wide variety of samples, including hard or abrasive materials. |
| Bovine Serum Albumin (BSA) | A well-characterized protein used as a standard for developing and validating LOD protocols in FTIR imaging [57]. |
| Microarray Printer | Used to fabricate standardized sample spots of precise volume and concentration for systematic LOD studies and calibration [57]. |
| Potassium Bromide (KBr) | Used in traditional transmission FTIR for preparing pellets of solid samples, allowing for bulk analysis. |
| Gold-Coated Glass Slides | Used in specialized reflection-absorption setups to achieve extreme sensitivity for detecting trace deposits, such as haze residues, with detection limits as low as 1 ng [58]. |
| Chemometric Software | Software equipped with Principal Component Analysis (PCA) and other multivariate tools to deconvolute complex spectra from mixtures and improve identification of unknowns [8] [59]. |
A deep and practical understanding of the detection thresholds and surface sensitivity of FTIR spectroscopy is not a limitation but a foundation for its rigorous application in research. By quantitatively acknowledging these constraints—such as the ~0.1-1.0 µm sampling depth of ATR and the statistical nature of the LOD—scientists can design better experiments, select the most appropriate sampling techniques, and interpret spectral data with greater confidence. The ongoing integration of advanced data analysis, including artificial intelligence and chemometrics, continues to push these boundaries, further solidifying FTIR's role as an indispensable tool in the identification of organic pollutants and the advancement of drug development.
Fourier Transform Infrared (FTIR) spectroscopy has established itself as a powerful analytical technique for identifying and characterizing organic pollutants across various environmental matrices. The fundamental principle underpinning this application is that molecular vibrations from chemical bonds within organic compounds absorb infrared light at specific frequencies, creating a unique vibrational fingerprint that can be used for identification [2]. However, the raw spectral data obtained from FTIR instruments is rarely pristine; it is often contaminated by various interfering factors such as light scattering, reflection, and optical interference, which manifest as baseline distortions, band shifts, and intensity variations [60]. These artifacts can obscure the crucial absorption information of the pollutant molecules, complicating both qualitative identification and quantitative analysis.
Within the context of a broader thesis on FTIR identification of organic pollutants, mastering data processing strategies becomes paramount. The successful translation of a complex, raw interferogram into a chemically meaningful spectrum that accurately represents the pollutant's composition relies heavily on robust data processing protocols. These protocols ensure that the identified spectral features genuinely correspond to the molecular vibrations of the pollutants rather than experimental artifacts or matrix effects. This technical guide provides an in-depth examination of two cornerstone data processing techniques—baseline correction and spectral deconvolution—detailing their fundamental principles, methodological approaches, and application within the specific domain of organic pollutant research. The ultimate goal is to equip researchers with the knowledge to enhance spectral quality, improve resolution, and extract reliable, reproducible chemical information from environmentally relevant samples.
Theoretical Foundations of FTIR Spectral Artifacts
An FTIR spectrum is a composite signal containing contributions not only from the fundamental absorbance of molecular bonds but also from various physical phenomena and instrumental effects. In environmental analysis, samples like water, soil extracts, or particulate matter can introduce significant light scattering due to suspended particles or heterogeneous surfaces [60]. This scattering effect causes a sloping baseline, which violates the fundamental assumption of the Beer-Lambert law that forms the basis for quantitative analysis. Furthermore, organic pollutants are frequently present in complex mixtures, leading to overlapping absorption bands. This overlap occurs when vibrational modes of different compounds, or different functional groups within the same molecule, absorb energy at similar frequencies, resulting in broad, poorly resolved spectral features that hinder accurate identification and quantification [61].
The process of converting a raw interferogram into a spectrum via the Fourier transform is a critical step that presupposes an ideal measurement conditions [2]. Real-world analyses of environmental samples often deviate from these ideals, introducing additional noise and artifacts. The figure below illustrates the journey from a raw, artifact-laden spectrum to a processed spectrum ready for analysis, highlighting the specific points where baseline correction and deconvolution intervene.
Baseline Correction: Methodologies and Protocols
Principles and Purpose
Baseline correction is a fundamental pre-processing step aimed at removing artificial spectral trends that are not attributable to the absorbance of infrared light by the sample's molecular bonds. These trends, or baselines, can arise from Mie scattering from large particles, reflectance from rough sample surfaces, or instrumental drift [60]. An uncorrected baseline negatively impacts subsequent analysis by distorting peak shapes, altering intensity measurements, and complicating the application of quantitative models. Effective baseline correction is therefore a prerequisite for obtaining accurate and reliable data, particularly when analyzing trace levels of organic pollutants in challenging environmental matrices.
Common Algorithms and Their Applications
Several algorithms have been developed for baseline correction, each with distinct strengths and ideal use cases. The choice of algorithm depends on the nature of the baseline artifact and the complexity of the underlying spectral profile.
Table 1: Common Baseline Correction Algorithms in FTIR Analysis
| Algorithm | Underlying Principle | Best For | Key Parameters | Considerations for Organic Pollutant Analysis |
|---|---|---|---|---|
| Linear/Polynomial Fitting | Models the baseline as a simple line or low-order polynomial curve. | Spectra with simple, smooth, and slowly varying baselines. | Polynomial order. | Fast and simple, but may underfit complex baselines common in environmental samples. |
| Modified Multi-polynomial Fitting (ModPoly) | Iteratively fits a polynomial to the spectrum, ignoring points identified as peaks. | Spectra with moderate baseline curvature. | Polynomial order, number of iterations. | More robust than simple polynomial fitting; effective for many soil and water extract samples. |
| Iterative Least Squares (IRS) | Uses least-squares fitting with asymmetric weighting to suppress peak regions. | Spectra with strong, complex baselines and high signal-to-noise ratio. | Asymmetry parameter, smoothing constraint. | Highly effective but requires careful parameter tuning to avoid distorting real pollutant bands. |
| Rubberband Correction | Constructs a baseline from the convex hull of the spectrum. | Spectra with intense scattering effects (e.g., from microplastics or biological tissues). | Number of baseline points. | Excellent for highly irregular baselines; commonly used in ATR-FTIR of complex samples. |
Detailed Experimental Protocol for Baseline Correction
The following protocol outlines a standardized workflow for applying baseline correction to FTIR spectra of organic pollutants.
Protocol 1: Iterative Baseline Correction for Complex Environmental Samples
Objective: To remove non-linear baseline artifacts from an FTIR spectrum of an organic pollutant mixture extracted from a water or soil sample.
Materials and Software:
- FTIR spectrometer (e.g., equipped with ATR accessory for solid samples) [14].
- Software capable of advanced spectral processing (e.g., MATLAB with PLS_Toolbox, Python with SciPy and NumPy, or commercial FTIR software suites).
- Sample spectra data file (e.g., .CSV, .SPA, .DX).
Procedure:
- Data Import and Pre-Smoothing: Import the raw spectrum. If the spectrum has a low signal-to-noise ratio, apply a mild smoothing filter, such as a Savitzky-Golay filter (e.g., 2nd polynomial order, 9-15 point window), to reduce high-frequency noise without significantly distorting the spectral features [60].
- Algorithm Selection: Based on an initial visual inspection of the raw spectrum's baseline shape, select an appropriate algorithm. For complex baselines, the Iterative Least Squares (IRS) method is recommended.
- Parameter Optimization:
- Set the asymmetry parameter (p) to a value between 0.001 and 0.1. A lower value is more aggressive in defining peaks and is suitable for spectra with broad peaks.
- Set the smoothing parameter (λ) to a value between 10² and 10⁹. A higher value results in a smoother baseline.
- It is critical to optimize these parameters on a representative subset of spectra. The goal is a baseline that follows the spectrum's valleys without cutting into the absorption peaks.
- Application and Validation:
- Apply the correction to the entire dataset.
- Validate the result by visually inspecting the corrected spectrum to ensure the baseline is flat and centered around zero absorbance. Compare the corrected peaks against a library spectrum or a standard, if available, to check for any peak distortion introduced by the correction process.
- Documentation: Record all parameters used (algorithm, p-value, λ-value, etc.) in the laboratory notebook or metadata file to ensure the reproducibility of the analysis.
Spectral Deconvolution: Methodologies and Protocols
Principles and Purpose
Spectral deconvolution is an advanced processing technique used to resolve overlapping absorption bands in an FTIR spectrum. Many vibrational modes in complex organic molecules or mixtures absorb in similar spectral regions, creating broad, composite peaks. Deconvolution mathematically enhances the spectral resolution, allowing for the identification and quantification of individual components within these enveloping bands [61]. For researchers studying organic pollutants, this is invaluable for identifying specific compounds within a complex contaminant profile, such as differentiating between various phenolic compounds or polycyclic aromatic hydrocarbons (PAHs) whose signatures often overlap.
Common Techniques and Their Applications
The two primary approaches to spectral deconvolution are Fourier Self-Deconvolution (FSD) and curve-fitting, which includes Gaussian, Lorentzian, and Voigt profile fitting.
Table 2: Common Spectral Deconvolution Techniques in FTIR Analysis
| Technique | Underlying Principle | Primary Application | Key Parameters | Advantages & Limitations |
|---|---|---|---|---|
| Fourier Self-Deconvolution (FSD) | Applies a narrowing function in the Fourier domain to mathematically enhance the apparent resolution of the spectrum. | Estimating the number and position of underlying components in a broad envelope. | Bandwidth (FWHH), narrowing factor (K). | Advantage: Provides an estimate of hidden peaks. Limitation: Does not directly quantify component areas; can amplify noise. |
| Curve-Fitting (Gaussian/Lorentzian) | Fits a sum of individual peak profiles (e.g., Gaussian, Lorentzian) to the experimental spectrum using a least-squares algorithm. | Quantifying the area and width of individual components within a complex band. | Peak center, height, width, and shape for each component. | Advantage: Provides quantitative information on each component. Limitation: Requires initial guesses for parameters; can be subjective. |
Detailed Experimental Protocol for Spectral Deconvolution
This protocol describes a hybrid approach using FSD followed by curve-fitting, which is a common and effective strategy for analyzing complex pollutant mixtures.
Protocol 2: Resolving Overlapping Peaks in Organic Pollutant Spectra
Objective: To resolve and quantify the individual components contributing to the broad C=O stretching band (1850-1650 cm⁻¹) in a mixture of organic compounds.
Materials and Software:
- A baseline-corrected FTIR spectrum (from Protocol 1).
- Software with FSD and non-linear curve-fitting capabilities (e.g., Opus, GRAMS/AI, OriginPro, or Python with Lmfit library).
Procedure:
- Spectral Region Isolation: Isolate the spectral region of interest (e.g., 1850-1650 cm⁻¹ for C=O stretches) from the fully pre-processed spectrum.
- Fourier Self-Deconvolution (FSD - Scouting Step):
- Apply FSD to the isolated region. The key parameters are the full width at half height (FWHH) of the original bands and a narrowing factor (K).
- Start with a FWHH estimate of 15-25 cm⁻¹ for solid or liquid samples and a moderate K-factor (e.g., 2.5). Adjust these parameters iteratively until hidden shoulders or peaks become visually apparent without excessive noise amplification [60].
- The output of this step is not used for quantification but to inform the number of underlying components and their approximate positions for the next step.
- Curve-Fitting Setup:
- Based on the FSD result, decide on the number of component peaks (n) to fit.
- Choose a peak profile. Lorentzian functions are often a good initial choice for IR bands, but a Voigt profile (a convolution of Gaussian and Lorentzian) can sometimes provide a better fit.
- Provide initial guesses for the center position, height, and width for each of the n components.
- Iterative Fitting and Validation:
- Run the non-linear least-squares fitting algorithm to optimize the parameters for all components simultaneously.
- Assess the quality of the fit by examining the residuals (difference between the experimental data and the fitted curve). A good fit will have residuals that appear as random noise centered around zero.
- The coefficient of determination (R²) should be >0.995 for a high-quality fit.
- The output provides the integrated area under each component peak, which is proportional to the concentration of the specific functional group or compound.
- Interpretation and Reporting: Assign the resolved peaks to specific molecular vibrations or pollutants based on their wavenumber position (e.g., 1740 cm⁻¹ for ester C=O, 1715 cm⁻¹ for carboxylic acid C=O, 1690 cm⁻¹ for amide I). Report the peak areas and centers for quantitative comparison.
The following diagram illustrates this multi-step workflow for spectral deconvolution, from the initial complex band to the final quantified components.
The Scientist's Toolkit: Essential Reagents and Materials
The successful application of the data processing strategies described above relies on a foundation of high-quality experimental data. The following table lists key reagents, materials, and software solutions essential for FTIR-based analysis of organic pollutants.
Table 3: Essential Research Reagents and Solutions for FTIR Analysis of Organic Pollutants
| Item Name | Function/Application | Technical Specification & Notes |
|---|---|---|
| ATR Crystal (Diamond/ZnSe) | Enables direct analysis of solid and liquid samples with minimal preparation via Attenuated Total Reflectance. | Diamond is durable and chemically inert, ideal for heterogeneous environmental samples. ZnSe offers a good balance of performance and cost but is softer and can be damaged. |
| IR-Grade Solvents (e.g., CHCl₃, CS₂, ACN) | Used for extracting organic pollutants from solid matrices (soil, sludge) and for preparing standard solutions for transmission cell analysis. | Must be anhydrous and spectroscopically pure to avoid introducing interfering absorption bands in the spectrum. |
| FTIR Spectrometer | The core instrument for collecting infrared absorption data. | Should be equipped with a DTGS or MCT detector. Portable FTIRs are emerging for field analysis of pollutants [14] [62]. |
| Software with Chemometrics | For advanced data processing, multivariate analysis, and machine learning. | Capabilities should include Principal Component Analysis (PCA), Partial Least Squares (PLS) regression, and the algorithms for baseline correction and deconvolution detailed in this guide [14] [60]. |
| Pellet Die Set | For preparing KBr pellets for transmission measurements of solid samples. | Allows for analysis of powders. Requires a hydraulic press. The process is more time-consuming than ATR but can provide superior spectra for some materials. |
| Certified Reference Materials (CRMs) | Pure organic pollutant standards for spectrometer calibration, library development, and method validation. | Critical for confirming the identity of peaks in unknown samples and for developing quantitative models. Examples include certified PAHs, pesticides, and plasticizers. |
Validating FTIR Analysis: Cross-Technique Comparison and Data Integrity
The identification and quantification of organic pollutants in environmental and biological matrices is a cornerstone of modern analytical chemistry, critical for public health protection and environmental monitoring. Within this field, Fourier Transform Infrared (FTIR) spectroscopy has emerged as a powerful tool for the preliminary characterization and identification of unknown organic materials and functional groups. However, no single analytical technique provides a complete picture. This whitepaper provides an in-depth technical comparison of FTIR against three cornerstone techniques: Atomic Absorption Spectroscopy (AAS), Inductively Coupled Plasma Mass Spectrometry (ICP-MS), and Gas Chromatography-Mass Spectrometry (GC-MS). Framed within the context of organic pollutant research, this guide elucidates the distinct roles, synergies, and optimal applications of each technique, empowering researchers and drug development professionals to design more effective and comprehensive analytical strategies.
Fundamental Principles and Comparative Analysis
Core Operational Principles
FTIR Spectroscopy: FTIR operates by passing infrared radiation through a sample and measuring the absorbance of specific wavelengths that correspond to the vibrational energies of molecular bonds. The resulting spectrum is a molecular "fingerprint" based on functional groups (e.g., C=O, N-H, C-O) that have a permanent dipole moment [52] [14] [63]. It is exceptionally versatile for identifying the "chemical family" of an unknown organic or polymeric substance [56].
Atomic Absorption Spectroscopy (AAS): AAS quantifies specific metallic elements by exploiting the phenomenon where ground-state atoms in a gaseous state absorb light at characteristic wavelengths. The amount of light absorbed is proportional to the concentration of the element in the sample [8].
Inductively Coupled Plasma Mass Spectrometry (ICP-MS): ICP-MS uses a high-temperature argon plasma (~6000-10,000 K) to atomize and ionize the sample. The resulting ions are then separated and quantified based on their mass-to-charge ratio (m/z). It is a premier technique for multi-element trace metal analysis [8].
Gas Chromatography-Mass Spectrometry (GC-MS): This hybrid technique first separates the individual components of a complex mixture using gas chromatography. Each separated component is then fragmented and identified by a mass spectrometer, providing both retention time and a unique mass spectrum for highly confident molecular identification [64].
Head-to-Head Technical Comparison
Table 1: Comparative analysis of FTIR, AAS, ICP-MS, and GC-MS for analytical applications.
| Parameter | FTIR | AAS | ICP-MS | GC-MS |
|---|---|---|---|---|
| Primary Analytical Target | Molecular functional groups & chemical bonds [56] | Specific metal elements [8] | Metal & non-metal elements; isotopes [8] | Organic molecules, volatile/semi-volatile compounds [64] |
| Detection Limits | ~1-10 wt% for quantification [56] | Parts per billion (ppb) to low parts per million (ppm) range [8] | Parts per trillion (ppt) to ppb [8] | Parts per billion (ppb) or lower [65] [64] |
| Quantitative Capabilities | Possible with standards and uniform thickness [56] | Excellent | Excellent | Excellent |
| Sample Throughput | High (rapid, non-destructive) [11] [14] | Moderate | High | Low to moderate (due to separation step) |
| Key Strengths | Rapid ID of organic functional groups, non-destructive, minimal prep [14] [56] | Cost-effective for specific metals, robust | Ultra-trace detection, multi-element analysis, wide dynamic range | Powerful separation, definitive ID of complex organics |
| Major Limitations | Poor sensitivity for metals, cannot detect simple ions [56] | Single-element analysis, limited dynamic range | High cost, spectral interferences | Limited to volatile/thermostable compounds, sample derivatization often needed |
Synergies in Organic Pollutant Research
In organic pollutant analysis, FTIR and GC-MS play interconnected yet distinct roles. FTIR serves as an excellent screening and preliminary characterization tool. For instance, in analyzing indoor dust for contaminants of emerging concern (CECs), Attenuated Total Reflection (ATR)-FTIR can rapidly separate organic compounds based on their functional groups from a complex mixture [66]. However, FTIR "falls short in molecular-level identification of CECs" [66]. This is where GC-MS becomes indispensable. The spectroscopic data from FTIR can be used to support the data obtained from the non-target analysis performed by GC-MS, leading to more confident identification of compounds like plasticizers, pharmaceuticals, and pesticides [66]. Similarly, FTIR is adept at characterizing the extraction materials and mechanisms used for isolating organic pollutants, while GC-MS confirms the specific identity of the extracted compounds [52].
For research involving toxic metals within organic matrices (e.g., metal-containing pesticides or organometallic compounds), FTIR's role shifts. While it cannot detect the metal itself, it can identify the functional groups and organic ligands bound to it [8] [63]. The quantitative profiling of the toxic metal is then performed by AAS or, for ultra-trace levels, ICP-MS [8]. This multi-technique approach provides a complete picture of the pollutant's structure and concentration.
Visualizing Method Selection and Workflow
The following diagram illustrates the decision-making pathway for selecting the appropriate analytical technique based on the research goal and analyte type, particularly in the context of organic pollutant analysis.
Experimental Protocols in Practice
Protocol 1: Identification of Organic Contaminants in Indoor Dust using ATR-FTIR and LC-Q-ToF-MS
This protocol demonstrates the synergistic use of FTIR and mass spectrometry for non-target analysis [66].
- Step 1: Sample Collection. Collect settled dust samples (e.g., ~100 g) from the micro-environments of interest (e.g., photocopying centers, residences) using a gentle sweeping motion and transfer to paper bags.
- Step 2: ATR-FTIR Analysis. Place the quartz fiber filter sample-side down directly onto the germanium crystal of the ATR-FTIR accessory. No extensive preparation is needed. Acquire spectra over the mid-infrared region (4000–400 cm⁻¹) with a resolution of 2 cm⁻¹, averaging 32 scans to improve the signal-to-noise ratio.
- Step 3: Spectral Interpretation. Identify functional groups and classes of compounds based on characteristic absorption peaks. For example, strong multiple C-O vibrations between 1300 and 900 cm⁻¹ may indicate the presence of anhydrides, while aliphatic hydrocarbons show C-H stretches near 2950, 2924, and 2850 cm⁻¹ [66].
- Step 4: LC-Q-ToF-MS Analysis. Extract the dust samples and analyze the extracts using Liquid Chromatography-Quadrupole Time-of-Flight Mass Spectrometry. This step provides precise molecular weight and fragmentation data for unambiguous identification.
- Step 5: Data Correlation. Use the functional group information from FTIR to support and confirm the molecular structures hypothesized from the LC-Q-ToF-MS data, creating a more robust analytical outcome [66].
Protocol 2: Analysis of Water-Soluble Components in Particulate Matter (PM)
This protocol highlights FTIR's application in environmental monitoring for identifying inorganic and organic components in air samples [37].
- Step 1: PM Sampling. Collect PM₁₀ and PM₂.₅ samples over a 24-hour period on 47 mm quartz fiber filters using a low-volume sampler operating at a constant flow rate (e.g., 2.3 m³/h).
- Step 2: Direct ATR-FTIR Analysis. Condition the filters in a controlled environment (T = 20 ± 1 °C, RH = 50 ± 5%) for 48 hours. Analyze the filter directly by placing it sample-side down on the ATR crystal, applying pressure to ensure good contact.
- Step 3: Identification of Ionic Species. Identify common water-soluble ions based on their unique vibrational frequencies:
- Sulfate (SO₄²⁻): 603, 615, 670, and 1100 cm⁻¹
- Ammonium (NH₄⁺): 1414 cm⁻¹
- Nitrate (NO₃⁻): 825 and 1356 cm⁻¹
- Carbonate (CO₃²⁻): 713, 730, and 877 cm⁻¹ [37]
- Step 4: Complementary IC Analysis. Validate and quantify the findings by analyzing water extracts of the filters using Ion Chromatography (IC), which is the standard method for determining soluble ion concentrations.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key reagents, materials, and instruments for FTIR-based analysis of organic pollutants.
| Item Name | Function/Application | Technical Notes |
|---|---|---|
| ATR Accessory | Enables direct, non-destructive analysis of solids, liquids, and multi-layer samples without extensive preparation [66] [56]. | Often features a diamond or germanium crystal. Allows for analysis of insoluble samples [56]. |
| Quartz Fiber Filters | Standard media for collecting particulate matter (PM) from air or dust samples for subsequent FTIR analysis [66] [37]. | Low background IR absorbance is critical for obtaining high-quality spectra of collected samples. |
| Bruker Tensor 27 Spectrometer | An example of an FTIR spectrometer used in research for acquiring high-quality infrared spectra [37]. | Typically operated with software like Bruker's OPUS for data collection and processing. |
| Chemometric Software | Advanced data processing using algorithms like Principal Component Analysis (PCA) and Partial Least Squares (PLS) [14]. | Extracts meaningful information from complex spectral data for classification and quantification. |
| Thermo Scientific Nicolet iS50 | A versatile FTIR spectrometer used for rapid identification of materials, including illicit drugs and trace evidence in forensic toxicology [11]. | Can be deployed with spectral search libraries for high-throughput screening. |
| Portable FTIR Spectrometer | Allows for on-site, real-time analysis in the field, such as at archaeological sites or for in-clinic medical diagnostics [11] [65] [14]. | Enables decentralized testing and rapid screening without the need for a central lab. |
The journey to definitively identify and quantify organic pollutants is not a solo endeavor for any single instrument. FTIR spectroscopy stands out as an unparalleled tool for rapid, non-destructive screening and functional group analysis, providing the first crucial clues about an unknown sample's chemical nature. However, as demonstrated, its true power is unlocked when used in concert with complementary techniques. GC-MS provides definitive molecular identification for volatile organics, while ICP-MS and AAS deliver ultra-trace quantification of elemental contaminants. The future of analytical research lies in the intelligent integration of these techniques, supported by advancements in chemometrics, portable instrumentation, and standardized protocols. By understanding the distinct advantages and limitations of each method detailed in this guide, researchers can construct more efficient, accurate, and comprehensive analytical workflows, ultimately advancing public health protection and environmental science.
Leveraging Spectral Libraries and Reference Databases for Verification
Fourier Transform Infrared (FTIR) spectroscopy has emerged as a powerful analytical technique for identifying organic pollutants based on their unique molecular fingerprints. This technical guide explores the critical role of spectral libraries and reference databases in verifying pollutant identities, ensuring analytical accuracy, and supporting regulatory compliance. We examine established methodologies, data processing protocols, and advanced verification workflows that leverage expanding spectral collections to address complex environmental matrices. The integration of these databases with chemometric analyses and complementary analytical techniques significantly enhances detection capabilities, enabling researchers to address evolving challenges in environmental monitoring, industrial safety, and public health protection.
Fourier Transform Infrared (FTIR) spectroscopy analyzes the interaction between infrared radiation and molecular structures, generating characteristic absorption patterns that serve as unique fingerprints for chemical identification [35]. When infrared radiation interacts with a sample, chemical bonds absorb energy at specific frequencies corresponding to their vibrational modes, producing a spectrum that reveals detailed information about functional groups and molecular structure [67]. This non-destructive technique provides rapid identification of organic compounds without extensive sample preparation, making it particularly valuable for environmental analysis where timely results are critical.
The identification of organic pollutants using FTIR spectroscopy relies on detecting characteristic absorption patterns associated with specific functional groups [10]. For instance, aromatic hydrocarbons like styrene display distinct out-of-plane bending of CH bonds in the aromatic ring around 910 cm⁻¹, while ketones such as acetone show broad absorptions centered at 1229 cm⁻¹ due to CC₂ antisymmetric stretching [10]. Similarly, alcohols including ethanol and isopropanol exhibit characteristic bands between 1010-1100 cm⁻¹ associated with various vibrational modes including wagging of CH₃ and antisymmetric stretching of CCO [10]. These spectral features enable researchers to not only identify but also discriminate between different environmental contaminants even in complex mixtures.
The application of FTIR spectroscopy in environmental monitoring has expanded significantly due to technological advancements that have improved sensitivity, resolution, and data processing capabilities [35]. Modern FTIR systems can detect volatile organic compounds (VOCs) at parts per million (ppm) levels using specialized configurations such as multipass gas cells that extend optical path lengths [10]. These developments, coupled with the technique's ability to provide real-time monitoring capabilities, have positioned FTIR spectroscopy as an indispensable tool for researchers addressing the growing challenges of organic pollution in various environmental compartments.
The Critical Role of Spectral Libraries in Pollutant Verification
Spectral Library Fundamentals
Spectral libraries function as comprehensive collections of reference spectra that serve as the cornerstone for accurate identification of organic pollutants through FTIR analysis [67]. The underlying principle of spectral matching operates on the concept that identical molecules produce identical infrared spectra, while different molecules yield distinct spectral patterns [67]. This fundamental characteristic enables researchers to compare unknown spectra against validated references to establish chemical identity through pattern recognition. The verification process employs specialized search software that calculates similarity metrics between the sample spectrum and library entries, typically presenting results as hit quality indices ranging from 0% (completely different) to 100% (identical) [67]. In practical applications, perfect matches are rare, and experienced analysts must visually confirm potential identifications, particularly for hits with quality indices below 70% that may still represent correct matches despite spectral variations caused by matrix effects or instrumental factors [67].
The value of any spectral library depends fundamentally on its quality and comprehensiveness. High-quality libraries provide accurate, well-annotated spectra collected under standardized conditions, while comprehensive collections cover diverse chemical classes relevant to environmental analysis. As noted in research on VOC detection, "The same molecules have the same infrared spectra, different molecules have different infrared spectra" [67], underscoring the foundational principle that makes library matching possible. However, successful identification requires that the target compound exists within the reference library, highlighting the importance of selecting libraries with appropriate scope and specialization for environmental pollutant analysis.
Types of Spectral Libraries
Spectral libraries vary significantly in their scope, specialization, and application focus. Understanding these variations is essential for selecting appropriate references for organic pollutant verification.
Table 1: Types of FTIR Spectral Libraries for Organic Pollutant Analysis
| Library Type | Scope and Coverage | Common Applications | Example Libraries |
|---|---|---|---|
| General Chemical | Broad collections of organic and inorganic compounds | Preliminary screening, unknown identification | Aldrich FTIR Library, Nicolet Condensed Phase Sampler Library [68] [67] |
| Specialized Environmental | Target pollutants, hazardous chemicals, VOCs | Environmental monitoring, regulatory compliance | Haz Chemicals Vapor Phase FTIR Library, Hydrocarbons FT-IR Library [68] |
| Commercial Databases | Comprehensive collections with 30,000+ spectra | Research laboratories, forensic analysis | Wiley Spectral Databases (1M+ IR spectra), Thermo Fisher Spectral Libraries [69] [68] |
| Custom/In-house | Laboratory-specific compounds, proprietary mixtures | Quality control, method development | User-generated libraries for specific applications [67] |
Recent expansions in commercial spectral libraries have significantly enhanced capabilities for organic pollutant identification. For instance, Wiley's spectral databases have grown to encompass more than 9.5 million spectra, including over 1 million IR spectra and 161,000 Raman spectra [69]. Similarly, specialized providers like NICODOM offer more than 140,000 infrared spectra across over 80 specialized libraries [67]. These expansive collections increase the likelihood of successful identification for emerging contaminants and complex transformation products that may be present in environmental samples.
Library Selection Criteria
Selecting appropriate spectral libraries requires careful consideration of several technical factors that directly impact verification accuracy. Spectral collection technique compatibility is paramount, as transmission spectra and ATR spectra exhibit systematic differences that can compromise matching accuracy if mixed [67]. With approximately 70-90% of new instruments deploying ATR sampling units, researchers must prioritize libraries collected with compatible techniques or apply mathematical corrections to address technique-specific variations [67]. The chemical relevance of a library to target analytes determines practical utility, with specialized collections often providing superior performance for specific pollutant classes compared to general libraries.
Additional considerations include spectral quality metrics such as signal-to-noise ratio, resolution, and appropriate background correction, along with metadata completeness containing detailed information about sampling conditions, instrument parameters, and compound provenance. For environmental applications focusing on volatile organic compounds, libraries containing vapor-phase spectra may be essential, as noted in VOC monitoring research: "The PID sensor (TA-2100 Styrene Detector from Mil-Ram Technology, Inc.) was calibrated for the detection of styrene in the range 1–100 ppmv" [10], highlighting the importance of context-specific calibration references. Regularly updated libraries that incorporate emerging pollutants and transformation products offer significant advantages for environmental applications where contaminant profiles continually evolve.
Experimental Protocols for Library-Based Verification
Sample Preparation and Handling
Proper sample preparation is critical for generating high-quality FTIR spectra that enable reliable library matching. The specific protocols vary significantly based on sample matrix and aggregate state, requiring researchers to implement appropriate methods for their analytical needs.
Gaseous Samples: For VOC analysis in air, researchers employ specialized gas cells with extended path lengths to enhance sensitivity. In a documented VOC monitoring setup, "a GEMINI Mars Series Multi-Pass Gas Cells 2 L/10 M with a nominal path length of 10 m and a volume of 2 L" was utilized to achieve detection sensitivity at ppm levels [10]. The protocol involves transferring evaporated VOCs from a sealed chamber into the pre-evacuated multipass cell, ensuring equilibrium conditions before spectral acquisition. This approach effectively concentrated target analytes and minimized interferents, with concentrations verified using photoionization detectors calibrated for specific compounds [10].
Solid Samples: Microplastic identification typically employs attenuated total reflection (ATR) techniques that require minimal sample preparation. The non-destructive nature of ATR-FTIR preserves sample integrity for subsequent analyses while providing high-quality spectra suitable for library matching [70]. For deposited particulate matter, studies have implemented passive sampling approaches with particles freely descending onto collection surfaces followed by direct ATR analysis without additional processing [71].
Liquid Samples: Aquatic organic pollution analysis may require concentration techniques to detect trace-level contaminants. Liquid-liquid extraction with compatible solvents followed by evaporation concentrates target analytes, while filtration removes particulate interferents. For direct analysis, specialized liquid cells with controlled path lengths prevent signal saturation from solvent absorption.
Table 2: Standardized Sampling Protocols for Different Environmental Matrices
| Sample Matrix | Recommended Preparation Method | Key Technical Considerations | Quality Control Measures |
|---|---|---|---|
| Ambient Air | Multi-pass gas cell (10m path) | Pre-evacuate cell to few mbar; verify equilibrium with PID | Calibrate PID with correction factors (e.g., CF=2.75 acetone, CF=1 styrene) [10] |
| Water | Solid-phase extraction or liquid concentration | Select appropriate sorbents for target pollutants; control pH | Use internal standards to monitor extraction efficiency; blank correction |
| Soil/Sediment | Solvent extraction followed by evaporation | Choose solvents based on pollutant polarity; clean-up steps | Matrix spike recovery tests; reference material analysis |
| Biological Tissue | Homogenization followed by lipid removal | Lyophilize samples; implement defatting procedures | Process blanks; confirm no spectral artifacts from preparation |
Consistent sample preparation across all samples in an analytical batch is essential for generating comparable spectra. Researchers must document all preparation steps meticulously, as variations in technique can introduce spectral artifacts that complicate library matching and verification. As emphasized in microplastic research, FTIR's non-destructive character "requires very small sample sizes and produces rapid results" [70], but this advantage depends entirely on appropriate handling protocols that preserve spectral fidelity.
Instrumental Parameters and Data Acquisition
Standardized instrumental parameters ensure spectral consistency and facilitate reproducible library matching across different instruments and laboratories. The following protocols outline optimal conditions for environmental sample analysis:
Spectral Resolution: A nominal resolution of 2-4 cm⁻¹ provides optimal detail for organic pollutant identification while maintaining acceptable signal-to-noise ratios. Research demonstrates that "a nominal resolution of 2 cm⁻¹ in the 400–5000 cm⁻¹ frequency range" effectively captures characteristic absorption features for VOC identification [10].
Scan Accumulation: Averaging multiple scans significantly improves spectral quality by reducing random noise. Studies typically collect "64 scans" per spectrum, balancing acquisition time with signal enhancement [10]. For trace analysis, increasing to 128 scans may provide additional noise reduction when sample concentration permits extended analysis time.
Spectral Range: The mid-infrared region (400-4000 cm⁻¹) captures fundamental vibrational modes most useful for compound identification. Some applications may benefit from extended ranges into the near-infrared (4000-10000 cm⁻¹) for overtone and combination band analysis, particularly for complex biological matrices [72].
Apodization Function: The Happ-Genzel function provides a reasonable default for environmental applications, though different functions may optimize resolution or sensitivity for specific analyses.
Advanced applications increasingly combine FTIR with complementary techniques to address analytical challenges. As noted in food analysis research, "FTIR primarily reflects the fundamental vibrational modes of molecular groups, providing more chemical information about the sample material" [72], while FT-NIR captures overtone and combination bands that can enhance multivariate modeling. Such integrated approaches provide more comprehensive contaminant characterization, particularly for complex environmental mixtures.
Data Processing and Analysis Workflow
Raw FTIR spectra require careful processing before meaningful library matching can occur. The following workflow outlines essential steps for preparing spectral data:
Atmospheric Compensation: Subtract characteristic absorptions from atmospheric H₂O (3000-4000 cm⁻¹ and 1750-2100 cm⁻¹) and CO₂ (around 2400 cm⁻¹) that otherwise obscure analyte signals [10]. This is particularly critical for gaseous samples analyzed in open-path configurations.
Baseline Correction: Apply appropriate algorithms (e.g., concave rubberband correction, linear interpolation) to remove scattering effects and instrumental offsets that distort spectral line shapes. The specific approach should match sample characteristics, with more aggressive correction for highly scattering samples like microplastics [70].
Smoothing: Implement Savitzky-Golay filtering or similar approaches to enhance signal-to-noise ratios without significant loss of spectral features, particularly important for trace-level detection.
Normalization: Apply vector normalization or standard normal variate (SNV) transformation to minimize concentration-dependent effects and focus library matching on spectral shape rather than absolute intensity.
Following preprocessing, library search operations employ similarity metrics such as correlation coefficients or Euclidean distances to identify potential matches. Hit quality indices provide preliminary ranking, but visual inspection by experienced analysts remains essential for verification, particularly for complex environmental samples with potential interferents. As emphasized in spectral interpretation guidelines, "You must always do a visual comparison of the unknown spectrum with the library spectrum. It is you who must decide if the spectrum is identical, not the software" [67].
Figure 1: FTIR Spectral Verification Workflow for Organic Pollutants
Advanced Integration and Verification Techniques
Chemometric Integration with Spectral Libraries
The integration of chemometric methods with traditional spectral library matching significantly enhances verification capabilities for complex environmental samples. Multivariate statistical techniques including Principal Component Analysis (PCA), Partial Least Squares Regression (PLSR), and Hierarchical Cluster Analysis (HCA) extract meaningful patterns from spectral data that may not be apparent through simple library matching alone [72].
Research demonstrates the effectiveness of PLSR modeling for predicting volatile organic compound concentrations based on FTIR spectral features. In studies analyzing Gastrodia elata, "112 partial least squares discriminant analysis (PLSR) prediction models were constructed from the full-spectrum and feature-number spectrum datasets using NIR and FTIR spectroscopy strategies" [72]. These models achieved high predictive accuracy for key flavor compounds, providing a framework for environmental pollutant quantification. The integration of multiple preprocessing methods with feature selection techniques further optimized model performance, highlighting the importance of tailored chemometric approaches for specific analytical challenges.
Machine learning algorithms represent a further advancement in spectral verification, with supervised methods including Support Vector Machines (SVM) and Artificial Neural Networks (ANN) enabling automated classification of complex spectral patterns [35]. These approaches learn distinguishing features from training sets of reference spectra, creating decision boundaries that facilitate identification even with overlapping spectral features or baseline variations. As noted in analytical reviews, "Advanced data processing enables automated identification of contaminants, quantification of concentration levels, and differentiation between similar organic compounds even in complex matrices" [35].
Hybrid Analytical Approaches
Combining FTIR with complementary analytical techniques addresses inherent limitations of individual methods and provides enhanced verification confidence through orthogonal data. Gas chromatography coupled with FTIR (GC-FTIR) separates complex mixtures before spectral analysis, resolving compounds that would otherwise produce overlapping spectral features [72]. This approach has successfully predicted key volatile organic compounds in botanical samples, with researchers noting that "NIR and FTIR combined with GC-MS technology can accurately predict key VOCs" [72].
Thermogravimetric analysis coupled with FTIR (TGA-FTIR) monitors thermal decomposition products in real-time, providing additional characterization dimensions for complex environmental samples like microplastics [70]. Similarly, integrating FTIR with mass spectrometry (MS) combines molecular fingerprinting with precise mass identification, creating a powerful tool for unknown pollutant identification. As summarized in contaminant analysis reviews, "These hybrid approaches combine FTIR with gas chromatography, mass spectrometry, thermal analysis, or microscopy to enhance identification capabilities and overcome limitations of individual methods" [35].
The growing availability of portable FTIR systems enables field-based analysis that complements traditional laboratory techniques. These "compact instruments incorporate miniaturized optical components, robust sampling interfaces, and battery operation capabilities" [35], supporting rapid screening and on-site decision making. While portable systems typically offer reduced resolution compared to laboratory instruments, their deployment for preliminary analysis helps prioritize samples for more comprehensive laboratory characterization, optimizing resource allocation in environmental monitoring programs.
Quantitative Analysis Methods
While FTIR spectroscopy excels at qualitative identification, quantitative analysis requires careful method development and validation. The Beer-Lambert law establishes the fundamental relationship between absorption intensity and analyte concentration, but practical application faces challenges including matrix effects, non-linear absorption at higher concentrations, and spectral overlaps in complex mixtures [35].
Multivariate calibration methods including Partial Least Squares (PLS) and Principal Component Regression (PCR) effectively address these challenges by modeling the relationship between spectral variations and reference concentrations. Research demonstrates that "quantitative analysis of organic contaminants involves calibration techniques and mathematical models to determine contaminant concentrations from FTIR spectral data" [35], with methods including multivariate analysis and partial least squares regression proving particularly effective. These approaches incorporate information from multiple spectral points, minimizing interference from overlapping signals and matrix effects.
For routine analysis of specific target compounds, univariate calibration using characteristic absorption peaks provides a simpler quantitative approach. This method requires careful selection of analyte-specific bands without interferents, verification of linear concentration responses, and implementation of internal standards to correct for instrumental variations. In VOC monitoring studies, quantitative analysis employed integrated peak areas for compound-specific absorptions, with calibration curves demonstrating "good performance in the quantitative assessment of organic pollution, with a coefficient of determination (R²) of 0.79" [10] [73].
Essential Research Reagents and Materials
Successful FTIR analysis of organic pollutants requires appropriate selection of research reagents and materials tailored to specific sample matrices and analytical requirements.
Table 3: Essential Research Reagents and Materials for FTIR Analysis of Organic Pollutants
| Category | Specific Items | Technical Function | Application Notes |
|---|---|---|---|
| Calibration Standards | Styrene, acetone, ethanol, isopropanol [10] | Quantitative method development; instrument calibration | Use certified reference materials; prepare serial dilutions for calibration curves |
| Sampling Accessories | KBr windows, BaF₂ cells, ZnSe ATR crystals [67] | Sample presentation and interface with instrumentation | Select crystal material based on spectral range, hardness, and chemical compatibility |
| Gas Analysis Systems | Multipass gas cells (10m path) [10], evaporation chambers | Enhanced sensitivity for gaseous samples; controlled vaporization | Pre-evacuate cells to few mbar; verify equilibrium before analysis |
| Reference Libraries | Commercial databases (Wiley, Thermo Fisher) [69] [68], custom libraries | Spectral matching and compound identification | Ensure technique compatibility (transmission vs. ATR); regular updates for new pollutants |
| Sample Preparation | Solvents (HPLC grade), filtration units, extraction sorbents | Matrix simplification; analyte concentration | Select solvents with minimal IR absorption; implement clean-up to remove interferents |
| Validation Tools | Photoionization detectors [10], internal standards | Method verification; quantification accuracy | Use PID correction factors (e.g., CF=2.75 acetone, CF=1 styrene) for concentration measurements |
The selection of appropriate sampling materials requires careful consideration of spectral properties and chemical compatibility. As emphasized in technical guidelines, "KBr is the only material which has no absorption bands in region 400-4000 cm⁻¹" [67], but its extreme sensitivity to moisture necessitates careful handling. Alternative materials including ZnSe, BaF₂, and diamond provide specialized properties for challenging applications, with ATR crystals increasingly dominating modern implementations due to simplified operation and minimal sample preparation requirements.
Spectral libraries and reference databases serve as foundational elements for effective FTIR analysis of organic pollutants, transforming raw spectral data into confident chemical identifications. The expanding coverage of commercial spectral collections, coupled with advanced data processing techniques and hybrid analytical approaches, continues to enhance verification capabilities for increasingly complex environmental samples. As spectral libraries grow to encompass emerging contaminants and transformation products, their value in environmental monitoring, regulatory compliance, and source tracking will continue to increase.
Future developments in spectral verification will likely focus on artificial intelligence integration, expanded field-deployable solutions, and enhanced data sharing frameworks that facilitate collaborative library development. By leveraging these evolving resources and methodologies, researchers can address the growing challenges of organic pollution with increasing sophistication, ultimately supporting more effective environmental protection and public health preservation.
Integrating FTIR with SEM-EDS and XRD for Comprehensive Material Characterization
The accurate identification and characterization of organic pollutants in complex environmental matrices present a significant challenge in research and drug development. While individual analytical techniques provide valuable data, they often offer only a partial view of the sample's complete chemical and physical nature. Fourier Transform Infrared (FTIR) spectroscopy serves as a powerful tool for identifying organic functional groups and molecular structures in pollutants, but when integrated with Scanning Electron Microscopy with Energy Dispersive X-Ray Spectroscopy (SEM-EDS) and X-Ray Diffraction (XRD), it enables a multidimensional analytical approach. This integration provides a comprehensive picture that encompasses molecular speciation, elemental composition, and crystallographic structure [74] [8].
This technical guide explores the synergistic combination of these techniques, framed within broader thesis research on identifying organic pollutants. By leveraging their complementary strengths, researchers can overcome the limitations of individual methods, gaining unprecedented insights into pollutant identity, origin, transformation pathways, and potential interactions in environmental and biological systems. The following sections detail the fundamental principles, practical methodologies, and specific applications of this integrated approach, providing a structured framework for its implementation in advanced research settings.
Fundamental Principles of the Individual Techniques
Fourier Transform Infrared (FTIR) Spectroscopy
FTIR spectroscopy analyzes the interaction between infrared radiation and matter by measuring the absorption of light at wavelengths that correspond to the vibrational energies of molecular bonds. The resulting spectrum provides a unique molecular fingerprint that can identify specific organic compounds and functional groups based on characteristic absorption patterns. Key absorption regions include the range for O-H stretching (around 3434 cm⁻¹), C-H stretching (2855-2925 cm⁻¹), and carbonyl C=O stretching (1690-1810 cm⁻¹) [74] [3]. For organic pollutant research, FTIR is particularly valuable for identifying hydrocarbon contaminants, microplastics, and various organic functional groups that may participate in metal binding or environmental transformations [8] [35]. Advanced FTIR methods such as Attenuated Total Reflectance (ATR) enable analysis of minimal samples with little to no preparation, making the technique ideal for rapid screening and identification of organic contaminants in diverse matrices [3] [6].
Scanning Electron Microscopy with Energy Dispersive X-Ray Spectroscopy (SEM-EDS)
SEM-EDS combines high-resolution imaging with elemental analysis. SEM provides detailed morphological and topographical information at micro- to nano-scale resolutions, revealing surface structures, microporosities, and particle morphology [74] [75]. When the electron beam interacts with the sample, it emits characteristic X-rays that EDS detects to provide qualitative and semi-quantitative elemental composition of the analyzed area. This technique identifies and quantifies elements present in a sample, with typical applications including determining Ca/P ratios in biological samples [75] or quantifying elemental concentrations in archaeological artifacts to determine their provenance [74]. Unlike FTIR, EDS does not identify molecular structures or organic compounds directly, but it provides crucial complementary data on elemental constituents that can influence the interpretation of organic pollutant data.
X-Ray Diffraction (XRD)
XRD is a powerful technique for determining the crystallographic structure of materials. It operates on the principle of X-ray diffraction by crystal lattices, where X-rays interact with a crystalline sample and are diffracted at specific angles according to the arrangement of atoms within the crystal. The resulting diffraction pattern provides information about crystal phases, crystal orientation, lattice parameters, and crystallite size [74] [76]. In the context of organic pollutant research, XRD is particularly valuable for identifying crystalline phases in environmental samples, such as mineral components that may interact with pollutants. For instance, XRD can identify high-temperature phases in clay minerals like wollastonite, anorthite, diopside, and mullite, which helps in estimating firing temperatures of pottery artifacts [74]. Unlike EDS and FTIR, XRD provides information on a bulk scale, representing the average crystallographic structure of the entire sample rather than specific localized areas [76].
Table 1: Comparative Overview of FTIR, SEM-EDS, and XRD Techniques
| Technique | Primary Information Obtained | Sample Requirements | Detection Capabilities | Key Applications in Pollutant Research |
|---|---|---|---|---|
| FTIR | Molecular functional groups, chemical bonds, molecular structure | Solids, liquids, powders; minimal preparation for ATR | Organic functional groups, polymers, inorganic ions | Organic pollutant identification, microplastic characterization, degradation studies [3] [35] |
| SEM-EDS | Surface morphology, elemental composition (≥Z=3), elemental mapping | Solid, conductive surfaces (coating may be required) | Elements from boron upwards, semi-quantitative composition | Particle morphology, elemental distribution, contamination mapping [74] [75] |
| XRD | Crystalline phases, crystal structure, crystallite size, lattice parameters | Powdered solids, thin films | Crystalline materials (detection limit ~1-5%) | Mineral composition, crystallinity assessment, phase identification [74] [76] |
Synergistic Integration of FTIR, SEM-EDS, and XRD
The true power of these analytical techniques emerges when they are strategically combined to leverage their complementary strengths. While FTIR excels at identifying organic functional groups and molecular structures, it typically cannot determine elemental composition or crystalline phases. Similarly, SEM-EDS provides elemental data but lacks molecular specificity, and XRD identifies crystalline phases but offers limited information about amorphous components or organic materials. This inherent limitation of individual techniques creates the imperative for their integration.
A robust integrated approach begins with FTIR analysis to identify the organic functional groups present in a sample. For instance, in analyzing microplastic pollution, FTIR can identify the polymer type (e.g., polyethylene, polypropylene, polystyrene) based on characteristic absorption peaks and assess the degree of environmental weathering through calculated indexes such as the Carbonyl Index (CI), Hydroxyl Index (HI), and Carbon-Oxygen Index (COI) [3]. These indexes track the formation of oxygen-containing functional groups (e.g., carbonyls around 1690-1810 cm⁻¹ and hydroxyl groups from 3100-3700 cm⁻¹) that indicate polymer degradation [3].
Following FTIR characterization, SEM-EDS analysis provides crucial complementary data on the material's morphology and elemental composition. SEM imaging can reveal surface features, cracks, and degradation patterns correlated with the chemical changes identified by FTIR [74]. EDS elemental analysis can detect inorganic additives, fillers, or adsorbed metals that may be associated with the organic pollutants. For example, in analyzing archaeological artifacts, EDS has been used to quantify elemental oxides (e.g., SiO₂, Al₂O₃, Fe₂O₃, P₂O₅) to determine the provenance and composition of materials [74].
XRD analysis completes the picture by identifying the crystalline phases present in the sample. This is particularly valuable for distinguishing between different mineral forms that may be associated with pollutants or for identifying crystalline degradation products. In environmental samples, XRD can detect crystalline mineral particles that may serve as substrates for organic pollutant adsorption or transformation [74]. The combination of these three techniques enables researchers to develop a comprehensive understanding of complex environmental samples containing both organic and inorganic components.
Table 2: Signature Identification Capabilities of an Integrated Approach
| Pollutant/Matrix Type | FTIR Contributions | SEM-EDS Contributions | XRD Contributions | Integrated Interpretation |
|---|---|---|---|---|
| Weathered Microplastics in Soil | Polymer identification (PE, PP, PS), degradation indexes (CI, HI, COI) [3] | Surface erosion patterns, microcracks, elemental composition of adherent particles [4] | Crystalline additives (e.g., TiO₂, fillers), mineral contaminants | Correlation of chemical degradation with physical weathering and filler composition |
| Organic Contaminants on Electronic Assemblies | Identification of flux residues, oils, fibers, organic films [77] | Localization of residues, elemental composition of corrosion products | Identification of crystalline corrosion products (e.g., sulfates, chlorides) | Distinguishing organic from inorganic residues and their interaction |
| Archaeological Artifacts (Pottery) | Mineral identification (calcite, quartz), firing temperature estimation [74] | Elemental composition for provenance, microstructure analysis [74] | High-temperature phase identification (e.g., wollastonite, mullite), firing temperature [74] | Comprehensive understanding of material composition, manufacturing techniques, and provenance |
Experimental Protocols and Methodologies
Sample Preparation Protocols
Proper sample preparation is critical for obtaining reliable and reproducible results across all three techniques. The specific preparation methods vary depending on the sample type and analytical goals:
FTIR Sample Preparation: For organic contaminant analysis, FTIR requires minimal preparation, especially when using ATR accessories which enable direct measurement of solids and liquids. Powdered samples can be analyzed as potassium bromide (KBr) pellets, where 1-2 mg of sample is mixed with 200 mg of dry KBr powder and pressed under vacuum to form a transparent pellet [74] [4]. Liquid samples can be analyzed as thin films between salt plates or using liquid cells. For microplastic analysis, environmental samples often require filtration onto infrared-transparent filters followed by direct ATR-FTIR analysis of collected particles [3].
SEM-EDS Sample Preparation: Samples must be electrically conductive and compatible with high-vacuum conditions. Non-conductive materials require coating with a thin layer (typically 10-20 nm) of conductive material such as gold, platinum, or carbon [75]. Carbon coating is preferred for EDS analysis as it doesn't interfere with the detection of light elements. Solid samples are mounted on aluminum stubs using conductive carbon tape or epoxy. For cross-sectional analysis, samples may require embedding in resin followed by polishing to reveal internal structures.
XRD Sample Preparation: Ideal samples for XRD are homogeneous fine powders with particle sizes less than 10 μm. Bulk samples should be ground gently in a mortar and pestle to minimize introducing strain into the crystals, which can affect diffraction line broadening. The powdered sample is then packed into a sample holder to create a flat, uniform surface. For thin films or specific orientation studies, specialized mounts may be required to preserve preferred orientation [74].
Integrated Analytical Workflow for Organic Pollutant Characterization
A systematic approach to integrating FTIR, SEM-EDS, and XRD ensures comprehensive characterization of environmental samples containing organic pollutants:
Initial FTIR Screening: Begin with FTIR analysis to identify the major organic functional groups and classes of compounds present. This guides subsequent analyses by highlighting potentially relevant inorganic components that require elemental and crystallographic characterization [35].
SEM-EDS for Morphology and Elemental Composition: Based on FTIR results, select representative areas for SEM-EDS analysis. SEM provides high-resolution images of particle morphology, surface features, and heterogeneity. EDS delivers elemental composition of specific particles or areas of interest, including the distribution of key elements associated with organic pollutants [74] [4].
XRD for Crystalline Phase Identification: Use XRD to identify crystalline phases in the sample. This is particularly important for understanding mineral associations with organic pollutants and identifying crystalline degradation products or additives [74].
Data Integration and Interpretation: Correlate findings from all three techniques to develop a comprehensive understanding of the sample. For example, FTIR might identify polyethylene based on characteristic C-H stretching bands, SEM-EDS might reveal adherent inorganic particles containing silicon and aluminum, and XRD could identify these particles as clay minerals, suggesting possible sorptive interactions between the microplastic and mineral phases [4].
Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Integrated Characterization
| Item | Function/Purpose | Application Notes |
|---|---|---|
| Potassium Bromide (KBr) | Matrix for FTIR pellet preparation | High-purity, infrared-grade for minimal background interference [74] |
| Conductive Carbon Tape | Mounting samples for SEM-EDS | Provides electrical conductivity and secure mounting on SEM stubs |
| Sputter Coater with Gold/Palladium or Carbon | Coating non-conductive samples | Creates thin conductive layer for SEM imaging; carbon preferred for EDS of light elements |
| Certified Polymer Reference Materials | FTIR spectral libraries and quantification standards | Polyethylene, polypropylene, polystyrene for microplastic identification [3] |
| Silicon Wafer Substrates | Optimal substrate for particulate samples | Minimal background for both FTIR and SEM-EDS analysis |
| Microporous Filters (e.g., Anodisc) | Filtration of aqueous samples for microplastic collection | Minimal infrared absorption for direct FTIR analysis of collected particles |
| Certified Elemental Standards | Calibration of EDS system | Required for quantitative elemental analysis |
Advanced Applications in Organic Pollutant Research
Microplastic Pollution Assessment
The integration of FTIR, SEM-EDS, and XRD has proven particularly valuable in microplastic pollution research. FTIR spectroscopy serves as the primary tool for polymer identification, with characteristic absorption peaks allowing discrimination between common plastics such as polyethylene (PE), polypropylene (PP), and polystyrene (PS) [3] [4]. Furthermore, FTIR can assess the degree of environmental weathering through calculated indexes. Research has shown that the Carbonyl Index (CI) increases with enhancing residence time in the environment, indicating photo-oxidative degradation of polymers [3].
SEM-EDS complements FTIR data by revealing the surface morphology of microplastics, showing features such as cracks, fractures, and erosion patterns that result from environmental degradation [3]. EDS analysis can detect inorganic additives or adsorbed metals on plastic surfaces, which may influence their toxicity and environmental behavior. For instance, studies of freshwater microplastics have shown adherence of various inorganic particles that can be characterized by EDS [4].
XRD analysis provides information about crystalline additives or fillers in plastics, such as titanium dioxide (TiO₂) pigments, which can be identified by their characteristic diffraction patterns. This integrated approach enables comprehensive characterization of microplastics in environmental samples, providing insights into polymer type, degradation state, and associated inorganic contaminants [4].
Analysis of Electronic Assembly Contaminants
In electronic manufacturing, residual organic contaminants on printed circuit board assemblies (PCBAs) can cause serious reliability issues including electrochemical migration and corrosion. FTIR microscopy has emerged as a powerful technique for identifying localized organic residues, fluxes, and discoloration on electronic assemblies, with analysis capabilities for areas as small as 5 × 5 μm [77].
When FTIR identifies an unknown organic residue, SEM-EDS can characterize its morphology and elemental composition, particularly identifying ionic species that may promote corrosion. XRD can then identify any crystalline corrosion products that have formed as a result of interactions between organic residues and metallic components. This multi-technique approach enables comprehensive root cause analysis of failures in electronic assemblies [77].
Archaeological and Cultural Heritage Materials
While not directly related to organic pollutants, the analysis of archaeological artifacts demonstrates the powerful synergy of these techniques. Studies of archaeological pottery have utilized FTIR to identify mineral components and estimate firing temperatures based on the presence or absence of characteristic minerals like calcite and amorphous aluminosilicates [74]. SEM-EDS has provided elemental composition data for provenance studies, while XRD has identified high-temperature crystalline phases that form during firing, providing additional constraints on production technologies [74]. This approach can be adapted to study long-term interactions between organic pollutants and cultural heritage materials.
The integration of FTIR spectroscopy with SEM-EDS and XRD represents a powerful multidimensional approach for comprehensive material characterization in organic pollutant research. Each technique brings unique and complementary capabilities: FTIR identifies molecular functional groups and organic compounds, SEM-EDS provides elemental composition and morphological context, and XRD determines crystalline phases and structures. Together, they enable researchers to overcome the limitations of individual techniques and develop a more complete understanding of complex environmental samples.
This integrated approach finds applications across diverse fields, from microplastic pollution assessment to contamination analysis in electronic assemblies and beyond. The strategic combination of these techniques, following standardized protocols and workflows, provides robust analytical frameworks for identifying organic pollutants, understanding their transformations, and assessing their interactions with inorganic components in environmental systems. As analytical technologies continue to advance, with improvements in sensitivity, resolution, and data processing capabilities, this integrated approach will become increasingly valuable for addressing complex challenges in environmental science and pollutant research.
The Role of Chemometrics and Machine Learning in Model Validation
In the field of analytical chemistry, Fourier Transform Infrared (FTIR) spectroscopy has established itself as a powerful, non-destructive technique for identifying molecular structures through their vibrational fingerprints. Within environmental research, FTIR plays a crucial role in detecting and profiling organic pollutants across diverse matrices, including air, water, and soil [8] [78]. However, the complexity of spectral data generated by FTIR instrumentation necessitates advanced computational approaches for meaningful interpretation. This is where chemometrics—the application of mathematical and statistical methods to chemical data—converges with modern machine learning (ML) to transform raw spectral information into reliable, actionable insights.
The integration of ML with FTIR spectroscopy represents a paradigm shift in analytical capabilities, enabling researchers to move beyond simple qualitative identification to sophisticated quantitative prediction and classification. Yet, the predictive power of these models is entirely dependent on the rigor of validation strategies employed during their development. Proper validation ensures that models are not merely memorizing training data (overfitting) but are genuinely learning underlying patterns that generalize to new, unseen samples [79]. This technical guide explores the foundational principles, methodologies, and practical applications of chemometrics and ML in model validation, specifically within the context of FTIR-based identification of organic pollutants.
Theoretical Foundations
From Classical Chemometrics to Artificial Intelligence
The evolution of data analysis in spectroscopy has progressed from classical statistical methods to increasingly sophisticated AI-driven approaches:
- Classical Chemometrics: Traditional methods like Principal Component Analysis (PCA) and Partial Least Squares (PLS) regression have formed the backbone of spectral analysis for decades. These linear methods are valued for their interpretability and efficiency in identifying latent variables within multivariate data [80].
- Machine Learning (ML): As a subfield of artificial intelligence (AI), ML develops models capable of learning from data without explicit programming. In spectroscopy, ML algorithms identify complex structures and patterns, improving analytical performance as they process more data [80] [81].
- Deep Learning (DL): This specialized ML subset employs multi-layered neural networks capable of hierarchical feature extraction. Architectures like Convolutional Neural Networks (CNNs) can automatically extract relevant features from raw or minimally preprocessed spectral data [80].
Core Machine Learning Paradigms
ML methods in chemometrics are generally categorized into three distinct learning paradigms:
- Supervised Learning: Models are trained on labeled data to perform regression (predicting continuous values) or classification (assigning categorical labels). Examples include PLS-DA, Support Vector Machines (SVM), and Random Forest, commonly used for spectral quantification and sample classification [80].
- Unsupervised Learning: Algorithms discover latent structures in unlabeled data. Techniques like PCA and clustering are primarily used for exploratory spectral analysis and outlier detection [80].
- Reinforcement Learning: Though less common in spectroscopy, these algorithms learn optimal actions by maximizing cumulative rewards in dynamic environments and are being explored for adaptive calibration [80].
The Critical Importance of Model Validation
The Overfitting Challenge
Overfitting represents the most significant challenge in developing predictive models from spectroscopic data. It occurs when a model learns not only the underlying relationship between spectral features and target properties but also the noise and random variations specific to the training dataset. The consequence is a model that performs exceptionally well on training data but fails to generalize to new samples [79]. This is particularly problematic in applications with significant public health implications, such as monitoring toxic metals in food products [8] or identifying organic pollutants in air particulate matter [78].
Key Validation Strategies
Proper validation strategies are essential for producing reliable, generalizable models:
- Cross-Validation: This approach systematically partitions the available data into training and validation subsets multiple times, providing a robust estimate of model performance while maximizing data usage. KFold cross-validation is widely employed in spectroscopic applications [82].
- External Validation: The gold standard for assessing model generalizability involves testing the final model on a completely independent dataset that was not used during any stage of model development or tuning [81].
- Uncertainty Estimation: Advanced techniques like Quantile Regression Forest (QRF) not only provide predictions but also quantify prediction uncertainty, offering valuable information about confidence levels for individual predictions [83].
The diagram below illustrates the comprehensive workflow integrating model development and validation:
Machine Learning Algorithms in FTIR Spectroscopy
Algorithm Selection and Performance
The selection of appropriate ML algorithms depends on the specific analytical task, data characteristics, and required interpretability. Recent research has evaluated multiple algorithms across various FTIR applications:
Table 1: Performance Comparison of Machine Learning Algorithms for FTIR Spectroscopy
| Algorithm | Common Applications | Key Advantages | Validation Performance Notes |
|---|---|---|---|
| Support Vector Machine (SVM) | Authentication of Chinese herbs [84], gelatin cross-linking analysis [82] | Effective in high-dimensional spaces; robust to noise | Achieved 100% accuracy in classifying 53 RRCH species [84] |
| Random Forest (RF) | Soil and agricultural analysis [83] | Reduces overfitting; provides feature importance rankings | Offers prediction intervals and uncertainty estimates [83] |
| Partial Least Squares-Discriminant Analysis (PLS-DA) | Identification of Gastrodia elata variants [85] | Interpretable; works well with correlated variables | Performance varies significantly with preprocessing methods [85] |
| Quantile Regression Forest (QRF) | Soil property prediction [83] | Provides prediction uncertainty estimates | 90% prediction interval found to be suitably accurate [83] |
| Decision Trees (DT) | Gelatin classification [82] | Highly interpretable; fast prediction | Prone to overfitting without proper regularization [82] |
| Linear Discriminant Analysis (LDA) | Gelatin cross-linking analysis [82] | Computationally efficient; works well with separated classes | Requires careful feature selection for optimal performance [82] |
Deep Learning Architectures
Deep Neural Networks (DNNs), particularly Convolutional Neural Networks (CNNs), have demonstrated remarkable performance in spectral analysis. The ResNet architecture applied to FT-NIR and ATR-FTIR data for identifying Gastrodia elata variants achieved 100% accuracy across all sample types, outperforming traditional machine learning models [85]. These architectures automatically learn hierarchical feature representations from raw spectral data, reducing the need for extensive manual preprocessing and feature engineering.
Experimental Protocols and Methodologies
Standardized Workflow for Model Development
A robust, validated model requires a systematic approach to experimental design and execution. The following protocol outlines key stages for developing and validating chemometric models for FTIR analysis of organic pollutants:
Phase 1: Sample Preparation and Spectral Acquisition
- Sample Collection: Ensure representative sampling across expected variability. For environmental pollutants, this includes collecting from different locations, time points, and concentration ranges [78].
- FTIR Parameters: Standardize instrumental conditions. Typical settings include:
- Spectral range: 4000-500 cm⁻¹
- Resolution: 4 cm⁻¹
- Scans: 32-64 per spectrum
- Detector: DTGS or MCT depending on sensitivity requirements [82]
- Quality Control: Incorporate replicate measurements and quality control samples to assess instrumental stability and measurement reproducibility.
Phase 2: Data Preprocessing
Effective preprocessing is essential for extracting meaningful information while minimizing artifacts:
- Normalization: Standardize spectral intensity using methods like Standard Normal Variate (SNV) or vector normalization to correct for path length variations [82].
- Baseline Correction: Remove fluorescence background and scattering effects using polynomial fitting or specialized algorithms [82].
- Smoothing: Apply Savitzky-Golay filters or similar approaches to reduce high-frequency noise while preserving spectral features.
- Spectral Alignment: Correct for subtle wavelength shifts using correlation optimization or peak alignment algorithms.
Phase 3: Model Development and Validation
- Data Splitting: Partition data into training (~70-80%), validation (~10-15%), and test (~10-15%) sets using stratified sampling to maintain class distributions.
- Model Training: Implement multiple algorithms with appropriate regularization to prevent overfitting.
- Hyperparameter Tuning: Use systematic approaches like grid search or random search optimized via cross-validation [79].
- Validation: Employ k-fold cross-validation (typically k=5 or 10) followed by external validation with completely independent test sets [81].
Case Study: Validation in Gastrodia elata Identification
A comprehensive study on identifying variants and hybrids of Gastrodia elata Blume demonstrated rigorous validation practices. Researchers collected 394 samples across multiple Chinese provinces and employed FT-NIR, ATR-FTIR, and their fusion with both traditional ML (PLS-DA, GS-SVM) and deep learning (ResNet) approaches [85]. Key validation aspects included:
- Testing multiple preprocessing techniques for each spectral dataset
- Comparing unsupervised (PCA, t-SNE) and supervised approaches
- Evaluating model performance across different sample types and geographical origins
- The ResNet model achieved perfect classification (100% accuracy), demonstrating the power of properly validated deep learning approaches [85]
Essential Research Toolkit
Table 2: Essential Research Reagents and Materials for FTIR-Chemometrics Research
| Item | Function/Application | Technical Specifications |
|---|---|---|
| ATR-FTIR Spectrometer | Spectral acquisition of solid and liquid samples | Diamond ATR crystal; DTGS or MCT detector; 4 cm⁻¹ resolution [86] |
| FT-NIR Spectrometer | Analysis of bulk materials and scattering samples | Wavelength range 800-2500 nm; high-sensitivity InGaAs detector [85] |
| KBr Powder | Preparation of pellets for transmission measurements | FTIR grade; purified; hygroscopic storage required [82] |
| Background Reference Materials | Instrument calibration and background collection | Dry air for atmosphere; appropriate solvent blanks [86] |
| Chemometrics Software | Data preprocessing, model development, and validation | Python with scikit-learn, Pybaselines; commercial packages (OPUS, MATLAB) [82] [81] |
| Standard Reference Materials | Method validation and quality control | Certified samples with known composition for model calibration [8] |
Advanced Validation Techniques
Uncertainty Estimation in Predictive Models
While traditional validation focuses on accuracy metrics, advanced applications require quantification of prediction uncertainty. Quantile Regression Forest (QRF) represents a significant advancement by providing prediction intervals alongside point estimates [83]. This approach:
- Uses decision-tree ensembles to capture the full conditional distribution of predictions
- Offers sample-specific uncertainty estimates
- Identifies regions where model predictions are less reliable (e.g., near detection limits or in sparsely populated regions of the feature space)
- In soil and agricultural applications, QRF produced accurate predictions with reliable 90% prediction intervals, enabling more informed decision-making [83]
Data Fusion and Multimodal Integration
Combining multiple spectroscopic techniques enhances model robustness through complementary information. Research on Gastrodia elata demonstrated that fusing FT-NIR and ATR-FTIR spectral datasets improved classification performance compared to either technique alone [85]. The validation of such multimodal approaches requires:
- Proper scaling and alignment of different spectral ranges
- Validation of fused models against each individual technique
- Assessment of whether performance improvements justify increased complexity
The integration of chemometrics and machine learning with FTIR spectroscopy has fundamentally transformed analytical capabilities for identifying organic pollutants and other chemical species. However, this powerful synergy is only as reliable as the validation strategies underpinning it. Through rigorous application of cross-validation, external validation, uncertainty estimation, and multimodal verification, researchers can develop models that not only excel with training data but maintain robust performance in real-world applications. As FTIR instrumentation advances and machine learning algorithms grow more sophisticated, the principles of comprehensive model validation will remain the cornerstone of trustworthy analytical science, ensuring that predictions lead to accurate conclusions and appropriate actions in environmental monitoring, food safety, and public health protection.
Conclusion
FTIR spectroscopy stands as a versatile, non-destructive, and efficient cornerstone technique for the identification of organic pollutants. Its power lies in providing specific molecular fingerprints that reveal functional groups and chemical structures, which is crucial for understanding pollutant origin, behavior, and toxicity. While limitations in sensitivity and matrix effects exist, they are effectively mitigated through optimized methodologies and integration with complementary analytical techniques. The future of FTIR in environmental and biomedical research is bright, driven by advancements in portable field-deployable instruments, the increasing application of machine learning for automated spectral analysis and classification, and its growing role in regulatory and quality control frameworks for ensuring public health safety.
References
- 1. Fourier Transform Infrared Spectroscopy (FTIR) [https://www.rjleegroup.com/ftir]
- 2. Fourier transform infrared spectroscopic technique for analysis ... [https://pubs.rsc.org/en/content/articlehtml/2025/na/d5na00522a]
- 3. Fourier Transform Infrared Spectroscopy to Assess the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9961336/]
- 4. FTIR based assessment of microplastic contamination in ... [https://www.nature.com/articles/s41598-025-14507-w]
- 5. EPA Method 320-Measurement of Vapor Phase Organic ... [https://www.federalregister.gov/documents/2024/03/01/2024-04359/epa-method-320-measurement-of-vapor-phase-organic-and-inorganic-emissions-by-extractive-fourier]
- 6. ATR-FTIR characterization of organic functional groups ... [https://www.sciencedirect.com/science/article/abs/pii/S1352231008009953]
- 7. FTIR Applications | Thermo Fisher Scientific - ZA [https://www.thermofisher.com/za/en/home/industrial/spectroscopy-elemental-isotope-analysis/molecular-spectroscopy/fourier-transform-infrared-spectroscopy/applications.html]
- 8. Emerging roles of FTIR spectroscopy in toxic metal profiling [https://foodsafetyandrisk.biomedcentral.com/articles/10.1186/s40550-025-00119-9]
- 9. Applications of Fourier Transform-Infrared spectroscopy in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10703385/]
- 10. High Sensitivity Monitoring of VOCs in Air through FTIR ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9370991/]
- 11. FTIR Applications | Thermo Fisher Scientific - US [https://www.thermofisher.com/us/en/home/industrial/spectroscopy-elemental-isotope-analysis/molecular-spectroscopy/fourier-transform-infrared-spectroscopy/applications.html]
- 12. designed for the analysis of large microplastics [https://pubs.rsc.org/en/content/articlehtml/2025/va/d4va00400k]
- 13. FTIR Analysis Beginner's Guide: Interpreting Results [https://www.innovatechlabs.com/newsroom/1882/interpreting-analyzing-ftir-results/]
- 14. A Review of the Latest Research Applications Using FT-IR ... [https://www.spectroscopyonline.com/view/a-review-of-the-latest-research-applications-using-ft-ir-spectroscopy]
- 15. Interpreting Infrared Spectra [https://specac.com/theory-articles/interpreting-infra-red-spectroscopy/]
- 16. How to Interpret FTIR Results: A Beginner's Guide [https://www.azooptics.com/Article.aspx?ArticleID=2756]
- 17. A Meta-Analysis Indeed and Practical Approach [https://www.mdpi.com/2073-4441/16/22/3170]
- 18. A Process for Successful Infrared Spectral Interpretation [https://www.spectroscopyonline.com/view/process-successful-infrared-spectral-interpretation]
- 19. Research on Quantitative Analysis Method of Infrared ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12299426/]
- 20. Quantitative analysis of dried serum FTIR spectra based on ... [https://www.sciencedirect.com/science/article/abs/pii/S1386142524015932]
- 21. FT-IR Spectroscopy Mini-Tutorial: Principles, Practice, and ... [https://www.spectroscopyonline.com/view/ft-ir-spectroscopy-mini-tutorial-principles-practice-and-applications-across-disciplines]
- 22. How an FTIR Spectrometer Operates [https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Spectroscopy/Vibrational_Spectroscopy/Infrared_Spectroscopy/How_an_FTIR_Spectrometer_Operates]
- 23. Analysis of Mixtures by FT-IR: Spatial and Spectral ... [https://www.spectroscopyonline.com/view/analysis-mixtures-ft-ir-spatial-and-spectral-separation-complex-samples]
- 24. Exploring FTIR in Polymer Structure and Change [https://universallab.org/blog/exploring_ftir_in_polymer_structure_and_change]
- 25. FTIR Functional Group Database Table with Search [https://instanano.com/all/characterization/ftir/ftir-functional-group-search/]
- 26. Developing transferable and universal IR biomarkers for ... [https://www.nature.com/articles/s41598-025-05068-z]
- 27. Optimising sample preparation for FTIR-based microplastic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8141486/]
- 28. Assessment of Organic Pollutants Desorbed from Plastic Litter ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12389833/]
- 29. FTIR Analysis: The First Step in Product Contamination ... [https://www.innovatechlabs.com/newsroom/1898/ftir-analysis-product-contamination/]
- 30. FTIR: Transmission vs ATR spectroscopy | Animated Guides [https://specac.com/theory-articles/transmission-vs-atr-spectroscopy-animated-guides/]
- 31. Using FT-IR Spectroscopic Imaging in Both ATR and ... [https://www.spectroscopyonline.com/view/using-ft-ir-spectroscopic-imaging-in-both-atr-and-transmission-modes]
- 32. Sample Preparation for FTIR Analysis [https://www.drawellanalytical.com/sample-preparation-for-ftir-analysis-sample-types-and-prepare-methods/]
- 33. Quantitative Fourier Transform Infrared spectroscopic ... [https://www.sciencedirect.com/science/article/pii/S004565359800486X]
- 34. An FTIR and EA-IRMS Application to the Degradation ... [https://www.mdpi.com/2076-3417/13/19/10851]
- 35. Analyze FTIR Data to Determine Organic Contaminants [https://eureka.patsnap.com/report-analyze-ftir-data-to-determine-organic-contaminants]
- 36. Exploring Fourier Transform Infrared (FTIR) spectroscopy ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11182784/]
- 37. ATR–FTIR Spectral Analysis and Soluble Components of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6679192/]
- 38. Fast screening compositions of PM2.5 by ATR-FTIR [https://www.sciencedirect.com/science/article/abs/pii/S1001074217321836]
- 39. Fourier Transform Infrared Spectroscopy (FTIR) [https://www.eurofins.cn/en/materials-and-engineering/technology/ftir/]
- 40. Mid-Infrared Sensing of Organic Pollutants in Aqueous ... [https://www.mdpi.com/1424-8220/9/8/6232]
- 41. TGA-IR-GC-MS: A powerful evolved gas analysis technique [https://ptgeindhoven.nl/publications/tga-ir-gc-ms-a-powerful-evolved-gas-analysis-technique/]
- 42. Fourier transform infrared spectroscopy as a tracer of ... [https://www.sciencedirect.com/science/article/abs/pii/S0031018221004077]
- 43. Quantitative Infrared Database - the NIST WebBook [https://webbook.nist.gov/chemistry/quant-ir/]
- 44. The NIST Quantitative Infrared Database - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4877123/]
- 45. FTIR Moisture Interference: Mitigating Water Vapor Peaks [https://eureka.patsnap.com/article/ftir-moisture-interference-mitigating-water-vapor-peaks]
- 46. Guide to FT-IR Spectroscopy [https://www.bruker.com/en/products-and-solutions/infrared-and-raman/ft-ir-routine-spectrometer/what-is-ft-ir-spectroscopy.html]
- 47. VaporFit: an open-source software for accurate ... [https://pubs.rsc.org/en/content/articlelanding/2025/cp/d5cp01007a]
- 48. Sampling Techniques for FTIR Spectroscopy - JASCO [https://jascoinc.com/learning-center/theory/spectroscopy/fundamentals-ftir-spectroscopy/sampling/]
- 49. FT-IR Search Algorithm – Assessing the Quality of a Match [https://www.spectroscopyonline.com/view/ft-ir-search-algorithm-assessing-quality-match]
- 50. Optimize Resolution in FTIR for Clarity in Results [https://eureka.patsnap.com/report-optimize-resolution-in-ftir-for-clarity-in-results]
- 51. An evaluation of resolution, accuracy, and precision in FT- ... [https://www.sciencedirect.com/science/article/abs/pii/S138614252400711X]
- 52. Application of fourier transform infrared (FTIR) ... [https://www.sciencedirect.com/science/article/pii/S2772582024000214]
- 53. Optimizing setup of scan number in FTIR spectroscopy ... [https://www.nature.com/articles/s41598-021-92858-w]
- 54. FTIR accessory selection guide [https://www.shimadzu.com/an/products/molecular-spectroscopy/ftir/ftir-spectroscopy-consumables/ftir-accessory-selection-guide/index.html]
- 55. Assessing Non-Ideal Instrumental Effects in High ... [https://egusphere.copernicus.org/preprints/2025/egusphere-2025-2232/]
- 56. Fourier Transform Infrared Spectroscopy (FTIR) [https://www.eag.com/techniques/spectroscopy/fourier-transform-infrared-spectroscopy-ftir/]
- 57. On the Limit of Detection in Infrared Spectroscopic Imaging [https://pmc.ncbi.nlm.nih.gov/articles/PMC10539114/]
- 58. FTIR surface analysis for conservation | npj Heritage Science [https://www.nature.com/articles/s40494-020-0349-8]
- 59. Fourier-transform Infrared (FT-IR) spectroscopy fingerprints ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7170381/]
- 60. Exploring the Steps of Infrared (IR) Spectral Analysis [https://www.mdpi.com/1420-3049/28/19/6886]
- 61. UV–vis and FT-IR Analysis Supported by TD-DFT ... [https://www.sciencedirect.com/science/article/pii/S0022286025027292]
- 62. Fourier Transform Infrared (FTIR) Spectroscopy for ... [https://www.mdpi.com/2076-3417/11/16/7416]
- 63. Application of Fourier Transform Infrared (FTIR ... [https://www.mdpi.com/1420-3049/30/3/684]
- 64. MS and GC–MS Analytical Methods for On-Line Thermally ... [https://www.mdpi.com/2227-9040/13/7/258]
- 65. Integrating AI and advanced spectroscopic techniques for ... [https://www.sciencedirect.com/science/article/pii/S0924224424005260]
- 66. ATR-FTIR and LC-Q-ToF-MS analysis of indoor dust from ... [https://www.sciencedirect.com/science/article/abs/pii/S0048969721021367]
- 67. Infrared spectra. Free Download. IR Spectra Library [https://www.infrared-spectra.com/]
- 68. Spectral Libraries [https://www.thermofisher.com/search/browse/featured/us/en/80013285]
- 69. Wiley expands spectral databases with new IR, Raman, LC ... [https://www.knowledgespeak.com/news/wiley-expands-spectral-databases-with-new-ir-raman-lc-ms-and-smartspectra-data/]
- 70. Fourier Transform Infrared Spectroscopy: An analytical ... [https://www.sciencedirect.com/science/article/abs/pii/S1350449523005285]
- 71. Micro-Characterization of Indoor Deposited Particles Using ... [https://aaqr.org/articles/aaqr-23-09-ar-0236]
- 72. Prediction of critical volatile organic compounds and ... [https://www.sciencedirect.com/science/article/abs/pii/S0963996925020782]
- 73. Quantitative Estimation of Organic Pollution in Inland Water ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12074346/]
- 74. FT-IR, XRD, SEM-EDS, EDXRF and chemometric analyses ... [https://www.sciencedirect.com/science/article/abs/pii/S0969806X17310915]
- 75. SEM, EDS, XRD, FTIR and XPS analysis [https://pubmed.ncbi.nlm.nih.gov/25306144/]
- 76. Difference between EDS analysis and XRD analysis? [https://rockymountainlabs.com/difference-between-eds-analysis-and-xrd-analysis/]
- 77. FTIR Analysis for PCB Residues & Contamination [https://www.zestron.com/en/know-how/applications-analytic/ftir-microscopy-analyse-residues-pcb]
- 78. Analytical methods for determining organic compounds ... [https://www.sciencedirect.com/science/article/abs/pii/S0165993619304273]
- 79. The importance of choosing a proper validation strategy in ... [https://www.sciencedirect.com/science/article/abs/pii/S0003267025012322]
- 80. Recent Research in Chemometrics and AI for ... [https://www.spectroscopyonline.com/view/recent-research-in-chemometrics-and-ai-for-spectroscopy-part-i-foundations-definitions-and-the-integration-of-artificial-intelligence-in-chemometric-analysis]
- 81. A Review of Machine Learning Methods Recently Applied ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10301390/]
- 82. Versatile machine learning algorithms for FTIR spectroscopy [https://link.springer.com/article/10.1007/s43621-025-01146-4]
- 83. Smarter Spectroscopy With a New Machine Learning ... [https://www.spectroscopyonline.com/view/smarter-spectroscopy-with-a-new-machine-learning-approach-to-estimate-prediction-uncertainty]
- 84. Machine learning and chemometric methods for high- ... [https://pubmed.ncbi.nlm.nih.gov/40328017/]
- 85. FT-NIR and ATR-FTIR spectroscopy combined with ... [https://www.sciencedirect.com/science/article/abs/pii/S0026265X2502079X]
- 86. FTIR Applications | Thermo Fisher Scientific - SA [https://www.thermofisher.com/sa/en/home/industrial/spectroscopy-elemental-isotope-analysis/molecular-spectroscopy/fourier-transform-infrared-spectroscopy/applications.html]