Research Articles
Mastering Sample Preparation for UV-Vis Spectroscopy in Pharmaceutical QC: A Guide to Accuracy, Compliance, and Efficiency
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on optimizing sample preparation for UV-Vis spectroscopy in pharmaceutical quality control.
UV-Vis Spectroscopy for Drug Quantification: Principles, Methods, and Validation in Pharmaceutical Analysis
This article provides a comprehensive guide to Ultraviolet-Visible (UV-Vis) spectroscopy for drug quantification, tailored for researchers and pharmaceutical development professionals.
Chromophores in Active Pharmaceutical Ingredients: From Molecular Design to Analytical Applications
This article provides a comprehensive overview of the critical role chromophores play in the development and analysis of Active Pharmaceutical Ingredients (APIs).
UV-Vis Wavelength Range in Pharma Analysis: A Comprehensive Guide from Fundamentals to Advanced Applications
This article provides a complete resource for researchers and drug development professionals on implementing Ultraviolet-Visible (UV-Vis) spectroscopy in pharmaceutical analysis.
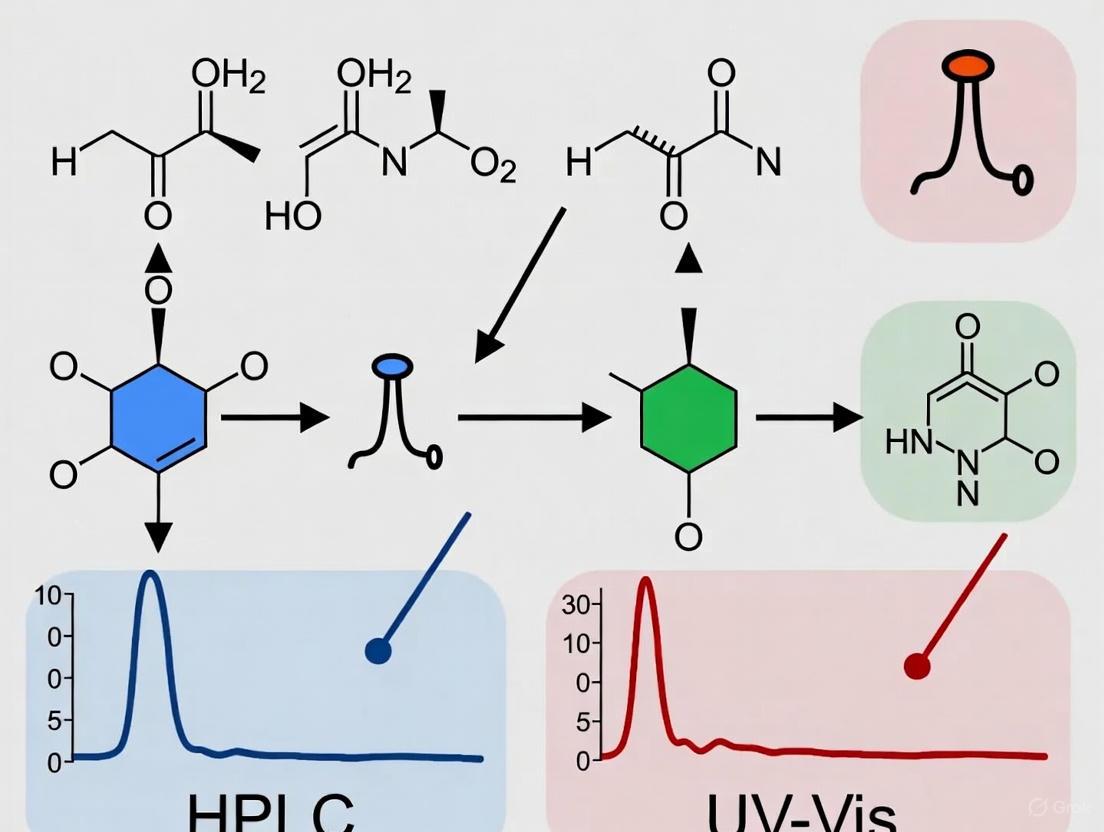
HPLC vs. UV-Vis: A Strategic Guide to Recovery Rate Comparison for Accurate Pharmaceutical Analysis
This article provides a comprehensive comparison of High-Performance Liquid Chromatography (HPLC) and UV-Visible Spectrophotometry (UV-Vis) methods, with a specific focus on recovery rates—a critical parameter for assessing analytical accuracy in...
Greenness in Analysis: A Comparative Evaluation of Fluorescence and Chromatographic Methods for Pharmaceutical and Bioanalytical Applications
This article provides a comprehensive comparative analysis of the environmental sustainability (greenness) of fluorescence-based and chromatographic analytical methods, a critical consideration for researchers and professionals in drug development.
Validation Parameters for Spectroscopic Methods: A Complete Guide for Researchers and Drug Development Professionals
This article provides a comprehensive guide to the validation of spectroscopic analytical methods, a critical process for ensuring data reliability in pharmaceutical development and biomedical research.
A Practical Guide to ICP-MS Validation Using Certified Reference Materials for Robust Analytical Results
This article provides a comprehensive framework for the validation of Inductively Coupled Plasma Mass Spectrometry (ICP-MS) methods using Certified Reference Materials (CRMs), specifically tailored for researchers and professionals in drug...
Infrared Spectroscopy in Food Analysis: A Comprehensive Guide to Quality Control and Authenticity Testing
This article provides a comprehensive overview of the application of infrared (IR) spectroscopy for ensuring food quality and authenticity.
Mitigating Spectral Artifacts in Handheld Raman Data: A Practical Guide for Robust Biomedical Analysis
Handheld Raman spectroscopy is revolutionizing pharmaceutical and biomedical analysis with its portability and non-destructive capabilities.